Abstract
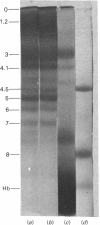
In addition to the microvesicles released during the treatment of human erythrocytes with Ca2+ and ionophore A23187, a new subpopulation of still smaller dense vesicles ('nanovesicles') has now been identified. Nanovesicles are about 60 nm in diameter, have an acetylcholinesterase activity higher than that of microvesicles and appear to be relatively enriched in sphingomyelin and correspondingly depleted of phosphatidylethanolamine. They have a polypeptide composition quite different from those of erythrocyte membranes or microvesicles, consisting largely of components of mol.wts. 60 000 and 26 000 in addition to haemoglobin. These two major polypeptides do not appear to represent contaminating cytoplasmic proteins or proteolytic subfragments of a larger protein.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allan D., Billah M. M., Finean J. B., Michell R. H. Release of diacylglycerol-enriched vesicles from erythrocytes with increased intracellular (Ca2+). Nature. 1976 May 6;261(5555):58–60. doi: 10.1038/261058a0. [DOI] [PubMed] [Google Scholar]
- Allan D., Michell R. H. A calcium-activated polyphosphoinositide phosphodiesterase in the plasma membrane of human and rabbit erythrocytes. Biochim Biophys Acta. 1978 Apr 4;508(2):277–286. doi: 10.1016/0005-2736(78)90330-9. [DOI] [PubMed] [Google Scholar]
- Allan D., Michell R. H. Accumulation of 1,2-diacylglycerol in the plasma membrane may lead to echinocyte transformation of erythrocytes. Nature. 1975 Nov 27;258(5533):348–349. doi: 10.1038/258348a0. [DOI] [PubMed] [Google Scholar]
- Allan D., Thomas P., Michell R. H. Rapid transbilayer diffusion of 1,2-diacylglycerol and its relevance to control of membrane curvature. Nature. 1978 Nov 16;276(5685):289–290. doi: 10.1038/276289a0. [DOI] [PubMed] [Google Scholar]
- Allen D. W., Cadman S. Calcium-induced erythrocyte membrane changes. The role of adsorption of cytosol proteins and proteases. Biochim Biophys Acta. 1979 Feb 20;551(1):1–9. doi: 10.1016/0005-2736(79)90348-1. [DOI] [PubMed] [Google Scholar]
- Allen D. W., Cadman S., McCann S. R., Finkel B. Increased membrane binding of erythrocyte catalase in hereditary spherocytosis and in metabolically stressed normal cells. Blood. 1977 Jan;49(1):113–123. [PubMed] [Google Scholar]
- BARTLETT G. R. Phosphorus assay in column chromatography. J Biol Chem. 1959 Mar;234(3):466–468. [PubMed] [Google Scholar]
- Beck J. S. Relations between membrane monolayers in some red cell shape transformations. J Theor Biol. 1978 Dec 21;75(4):487–501. doi: 10.1016/0022-5193(78)90358-2. [DOI] [PubMed] [Google Scholar]
- ELLMAN G. L., COURTNEY K. D., ANDRES V., Jr, FEATHER-STONE R. M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961 Jul;7:88–95. doi: 10.1016/0006-2952(61)90145-9. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lutz H. U., Liu S. C., Palek J. Release of spectrin-free vesicles from human erythrocytes during ATP depletion. I. Characterization of spectrin-free vesicles. J Cell Biol. 1977 Jun;73(3):548–560. doi: 10.1083/jcb.73.3.548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markwell M. A., Fox C. F. Surface-specific iodination of membrane proteins of viruses and eucaryotic cells using 1,3,4,6-tetrachloro-3alpha,6alpha-diphenylglycoluril. Biochemistry. 1978 Oct 31;17(22):4807–4817. doi: 10.1021/bi00615a031. [DOI] [PubMed] [Google Scholar]
- Palek J., Liu S. C., Snyder L. M. Metabolic dependence of protein arrangement in human erythrocyte membranes. I. Analysis of spectrin-rich complexes in ATP-depleted red cells. Blood. 1978 Mar;51(3):385–395. [PubMed] [Google Scholar]
- Quirk S. J., Ahkong Q. F., Botham G. M., Vos J., Lucy J. A. Membrane proteins in human erythrocytes during cell fusion induced by oleoylglycerol. Biochem J. 1978 Oct 15;176(1):159–167. doi: 10.1042/bj1760159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rumsby M. G., Trotter J., Allan D., Michell R. H. Recovery of membrane micro-vesicles from human erythrocytes stored for transfusion: a mechanism for the erythrocyte discocyte-to-spherocyte shape transformation. Biochem Soc Trans. 1977;5(1):126–128. doi: 10.1042/bst0050126. [DOI] [PubMed] [Google Scholar]
- Shukla S. D., Berriman J., Coleman R., Finean J. B., Michell R. H. Membrane protein segregation during release of microvesicles from human erythrocytes. FEBS Lett. 1978 Jun 15;90(2):289–292. doi: 10.1016/0014-5793(78)80388-3. [DOI] [PubMed] [Google Scholar]
- Skipski V. P., Peterson R. F., Barclay M. Quantitative analysis of phospholipids by thin-layer chromatography. Biochem J. 1964 Feb;90(2):374–378. doi: 10.1042/bj0900374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steck T. L. The organization of proteins in the human red blood cell membrane. A review. J Cell Biol. 1974 Jul;62(1):1–19. doi: 10.1083/jcb.62.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TAKAHARA S., HAMILTON H. B., NEEL J. V., KOBARA T. Y., OGURA Y., NISHIMURA E. T. Hypocatalasemia: a new genetic carrier state. J Clin Invest. 1960 Apr;39:610–619. doi: 10.1172/JCI104075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Zacharius R. M., Zell T. E., Morrison J. H., Woodlock J. J. Glycoprotein staining following electrophoresis on acrylamide gels. Anal Biochem. 1969 Jul;30(1):148–152. doi: 10.1016/0003-2697(69)90383-2. [DOI] [PubMed] [Google Scholar]