Abstract
In a murine model of acute fatal pneumonia, we previously showed that nasal immunization with a live-attenuated aroA deletant of P. aeruginosa strain PAO1 elicited LPS serogroup-specific protection, indicating that opsonic antibody to the LPS O antigen was the most important immune effector. Because P. aeruginosa strain PA14 possesses additional virulence factors, we hypothesized that a live-attenuated vaccine based on PA14 might elicit a broader array of immune effectors. Thus, an aroA deletant of PA14, denoted PA14ΔaroA, was constructed. PA14ΔaroA-immunized mice were protected against lethal pneumonia caused not only by the parental strain but also by cytotoxic variants of the O-antigen-heterologous P. aeruginosa strains PAO1 and PAO6a,d. Remarkably, serum from PA14ΔaroA-immunized mice had very low levels of opsonic activity against strain PAO1 and could not passively transfer protection, suggesting an antibody-independent mechanism was needed for the observed cross-serogroup protection. Compared with control mice, PA14ΔaroA-immunized mice had more rapid recruitment of neutrophils to the airways early after challenge. T cells isolated from P. aeruginosa ΔaroA-immunized mice proliferated and produced IL-17 in high quantities after co-culture with gentamicin-killed P. aeruginosa. Six hours following challenge, PA14ΔaroA-immunized mice had significantly higher levels of IL-17 in bronchoalveolar lavage fluid compared with unimmunized, E. coli-immunized, or PAO1ΔaroA-immunized mice. Antibody-mediated depletion of IL-17 prior to challenge or absence of the IL-17 receptor abrogated the PA14ΔaroA vaccine's protection against lethal pneumonia. These data show that IL-17 plays a critical role in antibody-independent vaccine-induced protection against LPS-heterologous strains of P. aeruginosa in the lung.
Keywords: Animals-Rodent, Infections-Bacterial, Molecules-Cytokines, Processes-Vaccination, Tissues-Lung
Introduction
Pseudomonas aeruginosa is a significant pathogen for hospitalized and immunocompromised patients. It is the most commonly isolated Gram-negative rod in the setting of hospital-acquired pneumonia (1) and, in the setting of ventilator-associated pneumonia, has a strikingly high attributable mortality (2). Vaccine development for lipopolysaccharide (LPS)-smooth strains of P. aeruginosa, which are the type of strains that cause nosocomial infections and are the initial colonizing strains in patients with cystic fibrosis (CF) (3), has been hampered by the inability to achieve broad protection across different LPS O antigen types (called serotypes or serogroups). In both mice (4) and humans (5), O antigen-based vaccine candidates have largely been abandoned due to lack of LPS-heterologous protection.
We are developing live-attenuated P. aeruginosa strains as intranasal vaccines and have shown previously that an unmarked aroA deletion mutant of the P. aeruginosa laboratory strain PAO1, denoted PAO1ΔaroA, is highly attenuated and, after intranasal immunization of mice, engenders high levels of serum opsonic antibody directed against LPS-homologous strains (6). Protection studies showed that after active immunization the PAO1ΔaroA vaccine prevented lethal pneumonia after inoculation with the parental strain PAO1, its cytotoxic variant ExoU+ PAO1, or some LPS-subtype related strains; but there was no protection after inoculation with LPS-heterologous strains (7). Passive immunization using antisera generated in nasally immunized rabbits protected only against lethal pneumonia due to ExoU+ PAO1 and failed to protect against LPS-heterologous strains or even against the parental strain PAO1 despite serum opsonic antibody titers that were identical to or higher than those observed after active immunization where protection was achieved (6). These results suggested both that the immunodominant nature of the LPS O antigen of PAO1ΔaroA may be hindering a broad antibody response and also that immune effectors elicited by active vaccination in addition to antibody are likely to be important for full protection against pneumonia.
Because of the broad host range of P. aeruginosa strain PA14 (8), we hypothesized that a live-attenuated vaccine based on this strain might elicit a broader array of immune effectors and thus constructed and tested an aroA deletion mutant in PA14 called PA14ΔaroA. Remarkably, we found that active intranasal immunization with PA14ΔaroA could protect against lethal pneumonia caused by LPS-heterologous strains in the absence of opsonic antibody. Further experiments showed that the neutrophil-attracting, T cell-secreted cytokine IL-17 is critical for the protective efficacy of this vaccine. Our results suggest that when opsonic antibody levels are low, rapid recruitment of neutrophils by IL-17-secreting vaccine-induced Th17 cells can achieve broad-based protections against P. aeruginosa pneumonia.
Materials and Methods
Bacterial strains
The bacterial strains and plasmids used in these experiments, along with their relevant characteristics and sources, are listed in Table I. PA14ΔaroA was constructed as previously described (6).
TABLE I.
Bacterial strains and plasmids used in this study
| Strain or plasmid | Description | Reference or source |
|---|---|---|
| P. aeruginosa | ||
| PAO1 | Wild-type strain, LPS-smooth, non-cytotoxic, serogroup O2/O5 |
M. Vasila |
| PAO1ΔaroA |
aroA deletion mutant of PAO1, LPS-smooth, serogroup O2/O5 |
(6) |
| PA14 | Wild-type strain, LPS-smooth, serogroup O10 | F. Ausubelb |
| PA14ΔaroA | aroA deletion mutant of PA14 | This study |
| ExoU+ PAO1 | PAO1(pUCP19exoUspcU). Cytotoxic variant of PAO1 carrying the plasmid pUCP19exoUspcU; Cbr |
(10) |
| PAO1 galU pUCP19 |
galU mutant of PAO1 in which the galU gene is disrupted by an aacC1 cassette; also carries the plasmid pUCP19; Gmr and Cbr |
(51) |
| ExoU+ PAO1 galU | PAO1 galU(pUCP19exoUspcU); Gmr and Cbr | (13) |
| ExoU+ PAO6a,d | PAO6a,d(pUCP19exoUspcU); Cbr | (10) |
| Fisher IT-7 | Wild-type strain, LPS-smooth, noncytotoxic, serogroup O2/O5 |
(4) |
| 6294 | Clinical isolate (corneal infection), LPS-smooth, noncytotoxic, serogroup O6 |
BPEIc, (52, 53) |
| 6206 | Clinical isolate (corneal infection), LPS-smooth, cytotoxic, serogroup O11 |
BPEIc, (52, 53) |
| PA103 | Clinical isolate, LPS-smooth, cytotoxic, serogroup O11 | D. Frankd |
| E. coli HB101 |
supE44 hsdS20(r−Bm−B) recA13 ara-14 proA2 lacY1 galK2 rpsL20 xyl-5 mtl-1 |
(54) |
| Plasmid | ||
| pUCP19 | Broad host range shuttle vector; Apr/Cbr | (55, 56) |
| pUCP19exoUspcU | pUCP19-based plasmid carrying the exoU gene and its chaperone gene, spcU |
(10) |
Abbreviations: Ap, ampicillin; Cb, carbenicillin; r, resistant
M. Vasil, Univ. of Colorado Health Sciences Center, Aurora, CO
F. Ausubel, Massachusetts General Hospital, Boston, MA
Bascom-Palmer Eye Institute, Miami, FL
D. Frank, Medical College of Wisconsin, Milwaukee, WI
Preparation of bacterial inocula for in vivo challenge studies
Frozen bacterial stocks were plated and grown overnight at 37°C on tryptic soy agar (TSA) or TSA containing the appropriate antibiotics (carbenicillin at 400 μg/mL, gentamicin at 150 μg/mL). For intranasal inoculation experiments, bacteria were suspended in PBS. For some survival experiments, bacteria were suspended in PBS containing 1% heat-inactivated fetal calf serum (FCS; Hyclone, Logan, UT). Concentrations were adjusted spectrophotometrically and confirmed after serially diluting in PBS containing 1% FCS and enumerating growth on TSA after overnight incubation at 37°C.
Immunization of mice
Six- to 8-week-old female C3H/HeN mice were purchased from Harlan Sprague-Dawley Farms (Chicago, IL) while C57BL/6 mice were purchased from Taconic (Hudson, NY). The C57BL/6 mice were age- and gender-matched with IL-17R knockout (KO) mice (also in C57BL/6 background) that were originally obtained from Amgen (Thousand Oaks, CA) and bred in-house. Mice were housed under virus-free conditions, and all animal experiments complied with institutional and federal guidelines regarding the use of animals in research. Immunizations were performed as described previously (6). Briefly, mice anesthetized with ketamine and xylazine were inoculated by placing 10 μL of the live P. aeruginosa ΔaroA vaccine strain or live E. coli HB101 as a control on each nostril. Escalating doses of 1 × 108 CFU, 5 × 108 CFU, and 1 × 109 CFU were administered at weekly intervals. For passive immunization experiments, mice were given pooled mouse antiserum (0.4 mL i.p.) 1 day prior to challenge. IL-17 depletion studies were done using IgG (1 mg i.p.) purified with protein G-linked agarose (Invitrogen, Carlsbad, CA) from an anti-mouse IL-17 rabbit antiserum kindly provided by A. Tzianabos (9). Control rabbit IgG was purchased from Sigma (St. Louis, MO).
Murine pneumonia model
All animal experiments complied with institutional and federal guidelines regarding the use of animals in research. We used our previously described model of acute fatal pneumonia following intranasal inoculation (10). In this model, 67-100% of the bacterial inoculum is consistently and rapidly translocated to the lungs from the nose of anesthetized 6- to 8-week-old mice (10) . For quantitation of CFU, neutrophils, and cytokines in bronchoalveolar lavage fluid (BALF), mice were sacrificed using CO2 at the indicated time points and then BALF obtained by cannulation of the trachea followed by two instillations of 1 mL of cold PBS containing 0.5 mM EDTA. For CFU measurements, BALF was diluted in 1% proteose peptone (BD, Franklin Lakes, NJ) and then plated on TSA for enumeration of CFU after overnight growth at 37°C. The limit of detection was 1 CFU in 100 μL of the undiluted BALF, which corresponded to 10 CFU per mL. BALF was centrifuged and the supernatant frozen at −80 °C prior to measurement of IL-17 by ELISA (R&D Systems, Minneapolis, MN). Erythrocytes in the cell pellets were lysed using a Mouse Erythrocyte Lyse Kit (R&D Systems) according to the manufacturer's instructions. Total cell counts were determined with a hemacytometer and Trypan Blue counterstaining. Percent neutrophils was measured by flow cytometry using PE-labeled anti-Ly6G (1A8) after treatment with anti-CD16/CD32 (2.4G2, FcBlock, BD Biosciences, San Jose, CA). For survival analyses, mice were followed daily for at least 7 days to assess mortality. Moribund animals were sacrificed and considered nonsurvivors.
Opsonophagocytic assays
Standard methods were employed, as described (6), with infant rabbit serum (Accurate Chemical, Westbury NY) as the complement source and polymorphonuclear leukocytes (PMNs) isolated from human volunteers. By convention, the serum concentrations listed were the input rather than final concentration (the initial serum dilution is further diluted 4-fold in the assay tube). Under routine conditions, killing of ≥50% is considered biologically significant and therefore serves to classify a serum as positive for opsonic killing activity. Although killing <50% is sometimes statistically significant, this level of killing is not considered biologically significant.
Anti-P. aeruginosa IgG titers
P. aeruginosa-specific IgG binding curves were measured by ELISAs using plates coated with whole live bacteria as previously described (6).
T cell proliferation, stimulation, and cytokine measurements
Splenic T cells were isolated using CD3 cell selection columns from R&D Systems. Cell purity was routinely >90% CD3+ cells as assessed by flow cytometry (not shown). Lung leukocytes were isolated after perfusion of the lungs in situ with PBS containing heparin (10 Units/mL) followed by incubation with EDTA, then digestion with collagenase and DNase, then sedimentation over a discontinuous Percoll gradient, all as previously reported (11). Bacteria used as antigens were killed by incubation of a 109 CFU/mL suspension with gentamicin (1 mg/mL in PBS) for 1 h at 37 °C, with killing verified by absence of growth of 100 μL samples plated on TSA. Staphylococcal enterotoxin A (SEA) was purchased from Sigma (St. Louis, MO) and used at final concentration of 10 ng/mL. For proliferation experiments, each well of a 96-well round-bottom plate was seeded with 1×105 T cells, 1×105 irradiated (1500 rad) splenocytes isolated from unimmunized C3H/HeN mice and purified using density centrifugation over Lympholyte-M (Accurate Chemical), and 1×106 gentamicin-killed bacteria, all suspended in RPMI containing 10% heat-inactivated FCS as well as standard concentrations of glutamine, sodium pyruvate, nonessential amino acids, penicillin, streptomycin, and 1 mM 2-mercaptoethanol (all from Invitrogen, Carlsbad, CA). On day 5 of stimulation, wells were pulsed with [3H]thymidine (1 μCi/well) for 8 hours prior to scintillation counting.
For cytokine secretion experiments, 96-well plates were used containing 1×105 T cells and 1×105 irradiated splenocytes as feeder cells. For both proliferation and cytokine secretion experiments, anti-CD4 (GK1.5), anti-CD8 (53-6.7), and rat IgG isotype controls were purchased from BD Biosciences and used at 1 μg per well. For stimulation of bulk lung leukocytes isolated 18 h after challenge with live bacteria, no feeder cells were added. IL-17 and IFN-γ were measured in supernatants after 7 days of stimulation using ELISA (R&D Systems). Intracellular cytokine staining was performed using a kit from BD Biosciences according to the manufacturers instructions. Lung leukocytes isolated as described above 2 h after challenge with live bacteria were stimulated for 15 h ex vivo with PMA and ionomycin (Leukocyte Activation Cocktail, BD Biosciences) in the presence of Brefeldin A (GolgiPlug, BD Biosciences). After treatment with anti-CD16/CD32, cells were stained with FITC-labeled antibodies to mouse TCRβ (H57-597) or CD3ε (145-2C11), followed by fixation and permeabilization and then staining with PE-labeled anti-mouse IL-17 (TC11-18H10.1) and analysis on a MoFlo (Dako, Carpinteria, CA) flow cytometer. Antibodies and appropriate isotype controls were obtained from BD Biosciences.
Statistical analyses
Nonparametric data were evaluated by Mann Whitney U test (for 2-group comparisons) or Kruskal Wallis (for 3 or more groups) with Dunn's multiple comparison test for pairwise comparisons. Parametric data were analyzed by t test (for 2-group comparisons) or ANOVA with Dunnett's multiple comparison test using E. coli-immunized mice or cells as the control comparator group. All analyses were performed using Prism software (GraphPad Software, San Diego, CA). Survival data were analyzed by Fisher's exact test or by survival analysis with the Kaplan-Meier method, also using Prism.
Results
Protective efficacy of PA14ΔaroA vaccination
We initially evaluated the protective efficacy of the PA14ΔaroA vaccine against LPS-homologous and heterologous strains. In our prior work, we showed that the serogroup O2/O5 vaccine PAO1ΔaroA elicited high levels of opsonic antibody as well as protection from lethality after lung challenge with LPS-homologous strains but not LPS-heterologous strains. As shown in Table II, nasal immunization with PA14ΔaroA resulted in significantly better survival compared with E. coli-immunized mice after challenge with a high dose of the parental strain PA14. There was no protection after challenge of PAO1ΔaroA-immunized mice with a lower dose of strain PA14. Notably, PA14ΔaroA-immunized mice were protected from lethality after challenge with ExoU-producing strains of the LPS-heterologous strains PAO1 (serogroup O2/O5) and PAO6a,d (serogroup O6). There was no protection, however, after challenge with the non-cytotoxic strains Fisher IT-7 (serogroup O2/O5) or 6294 (serogroup O6). This is likely related to the higher LD50s of the non-cytotoxic strains in this model and thus the very high challenge doses required to achieve 100% mortality in the E. coli-immunized control mice.
TABLE II.
Protective efficacy against acute fatal P. aeruginosa pneumonia following active nasal immunization of C3H/HeN mice with E. coli HB101, PAO1ΔaroA, or PA14ΔaroA
| Vaccine Group |
||||||
|---|---|---|---|---|---|---|
| Challenge Strain |
Sero- group |
LD50b (CFU) |
Inoculum (CFU) |
E. coli HB101 |
PAO1ΔaroA | PA14ΔaroA |
| ExoU+ PAO1 | O2/O5 | 7 × 105 | 2 × 107 | 1/7a | 7/8* | |
| PA14 | O10 | 3 × 106 | 1 × 108 | 0/4 | 0/4 | |
| 8 × 108 | 2/9 | 9/9** | ||||
| ExoU+ PAO6a,d | O6 | 1 × 106 | 2 × 107 | 1/5 | 10/10** | |
| 7 × 106 | 3/11 | 1/4 | ||||
| Fisher IT-7 | O2/O5 | 7 × 106 | 6 × 108 | 1/4 | 1/4 | |
| 6294 | O6 | 5 × 107 | 2 × 108 | 1/6 | 1/5 | |
| 6206 | O11 | 4 × 106 | 4 × 106 | 1/8 | 4/8# | |
| PA103 | O11 | 2 × 105 | 1 × 106 | 3/6 | 3/6 | |
Opsonic killing activity in antisera raised to P. aeruginosa PA14ΔaroA
For most vaccines effective against P. aeruginosa infections, serum opsonic antibody directed against the LPS O antigen has been the most important immune effector (12). To assess the role of serum antibodies and antibodies to specific antigens such as the ExoU cytotoxin in the protection afforded by the PA14ΔaroA vaccine, we first tested the opsonic killing activity of pooled sera obtained from immunized mice 3 weeks after the third (final) nasal immunization. As depicted in Fig. 1A, there was minimal opsonic killing of strain PAO1 in serum from mice immunized with the PA14ΔaroA strain, whereas good killing was achieved using sera from mice immunized with the LPS-homologous PAO1ΔaroA strain. Opsonic killing activity in antisera raised to PA14ΔaroA was present against the parental PA14 strain, although the opsonic titer (the calculated serum dilution at which the killing activity dropped to 50% as determined by linear regression) was approximately 50 (not shown), which is much lower than the titer of approximately 1000 seen with antiserum to PAO1ΔaroA against its parental strain PAO1 (7). Because PA14ΔaroA possesses the genes for ExoU while PAO1ΔaroA does not, and thus the PA14-based vaccine could induce antibody to this antigen, we next evaluated whether antibody to ExoU could be mediating opsonic killing if for unexpected reasons this antigen was retained on the bacterial cell surface. As shown in Fig 1B, there was no significant opsonic killing of an ExoU-producing strain compared with the strain containing the empty vector.
Fig. 1.

Absence of PAO1-targeted (A) and ExoU-specific (B) opsonic killing activity of sera from PA14ΔaroA-immunized mice 3 weeks after the final immunization. Bars are means and error bars the SEM. P values calculated by ANOVA with Tukey's multiple comparison test. C. Survival after passive immunization with PA14ΔaroA mouse antiserum (0.4 mL i.p., n=5 mice/group) given 1 day prior to lung challenge with P. aeruginosa strain ExoU+ PAO1 (1.4 × 107 CFU per mouse, a dose that resulted in 100% lethality of mice passively immunized with E. coli antiserum [not shown]).
Passive immunization and whole-cell ELISA experiments
Next, we determined whether passive immunization with pooled antisera from PA14ΔaroA-immunized mice mediated protection against lethal pneumonia due to the LPS-heterologous strain ExoU+ PAO1. Mice given the PA14ΔaroA antiserum i.p. 1 day prior to challenge had significantly higher mortality compared with the 0% mortality of mice given identical amounts of antisera raised to the LPS-homologous PAO1ΔaroA strain (Fig. 1C). These results suggested, surprisingly, that an antiserum-independent mechanism is a key component of the heterologous protection engendered by active vaccination with the PA14ΔaroA vaccine. We also tested for O antigen-specific and ExoU-specific antibodies by comparing IgG titers of the PAO1ΔaroA- and PA14ΔaroA-immunized mouse antisera to whole cells of the wild-type parental strain PAO1 (containing the empty vector) compared with strain ExoU+ PAO1 as well as ExoU-producing and non-producing LPS-rough galU mutants of strain PAO1 (galU mutants are O antigen deficient with a truncated outer LPS core (13)). As shown in Fig. 2, the PA14ΔaroA antisera had the expected lower reactivity than did the antisera raised to PAO1ΔaroA against P. aeruginosa strains PAO1 (pUCP19) and ExoU+ PAO1 based on its different LPS serogroup. The titers to the O antigen-deficient galU mutants were identical, suggesting that the PA14ΔaroA vaccine elicited a comparable level of antibody to non-O-side-chain antigens as did the PAO1ΔaroA vaccine.
Fig. 2.

Comparison of IgG binding curves to P. aeruginosa strains PAO1 (with empty vector pUCP19), ExoU+ PAO1, and their LPS-rough isogenic galU mutant cells by ELISA using antisera from C3H/HeN mice immunized with live E. coli, PAO1ΔaroA, or PA14ΔaroA bacteria. NMS: normal mouse serum. Each point is the mean of duplicates using pooled sera from 3-5 mice.
T cell responses elicited by P. aeruginosa PA14ΔaroA vaccine
To determine whether immune T cells are induced by nasal immunization with the P. aeruginosa ΔaroA vaccines, we isolated splenic T cells 3 weeks after immunization (prior to challenge) and co-cultured them with irradiated splenocytes as antigen-presenting cells (APCs) and gentamicin-killed P. aeruginosa as antigen and measured T cell proliferation by [3H]thymidine incorporation after 5 days. As shown in Fig. 3A, there was a high level of proliferation of both PAO1ΔaroA and PA14ΔaroA-immune T cells to either PAO1 or PA14, although the PA14ΔaroA-immune T cells proliferated less well when stimulated with PAO1 compared with the PAO1ΔaroA-immune T cells. Proliferation of PAO1ΔaroA-immune T cells was inhibited by an antibody to CD4 but not to CD8 (Fig. 3B). Proliferation of PAO1ΔaroA and PA14ΔaroA-immune T cells was high when stimulated with multiple P. aeruginosa strains regardless of LPS serogroup or ability to produce ExoU (Figs. 3C). Interestingly, the P. aeruginosa ΔaroA-immune T cells proliferated when cultured with E. coli but the E. coli-immune T cells were not significantly stimulated by P. aeruginosa strains.
Fig. 3.
Nasal immunization with P. aeruginosa ΔaroA vaccines primes splenic T cells. A. Proliferation of splenic T cells from unimmunized mice compared with mice immunized with E. coli, PAO1ΔaroA, or PA14ΔaroA. B. Proliferation of PAO1ΔaroA-immune T cells was significantly decreased by anti-CD4 monoclonal antibody. C. Proliferation of T cells from PAO1ΔaroA (left panel) or PA14ΔaroA (right panel) immunized mice was similar regardless of LPS serogroup or cytotoxic genotype of the stimulating gentamicin-killed, whole bacterial cell antigen (MOI: 10 bacteria per T cell). Bars represent means and error bars SEM. Cells were pooled from 3-5 mice per group. In A, *p<0.05 and #p<0.01 in comparison to E. coli control group by ANOVA with Dunnett's multiple comparison test. In B, *p<0.05 compared with IgG control of corresponding vaccine group and #p<0.01 by t test compared to E. coli-immune T cells. In C, proliferation of ΔaroA-immune T cells was significantly greater (p<0.01, t test) compared to E. coli-immune T cells in the presence of P. aeruginosa. SEA: staphylococcal enterotoxin A, positive control; n.d., not determined.
Cytokine secretion by P. aeruginosa ΔaroA vaccine-elicited T cells
As neutrophils are known to be essential mediators of protective immunity in the lung against P. aeruginosa infection (14, 15), we hypothesized that the recently described Th17 subset of CD4 helper T cells, which secrete the neutrophil-attracting cytokine IL-17, might play a role in rapid recruitment of neutrophils to the airways. We thus evaluated IL-17 secretion after 7-day co-culture of splenic T cells from naïve and immunized mice cultured with irradiated splenocytes and gentamicin-killed P. aeruginosa strains PAO1 and PA14 as antigens (Figs. 4A and 4B). For comparison, we also measured levels of IFN-γ, the predominant Th1 effector cytokine, and found them not to be significantly different among the vaccine groups. In contrast, the levels of IL-17 in the supernatants of the P. aeruginosa ΔaroA-immune T cells stimulated with P. aeruginosa were significantly higher than those of control T cells. Addition of the anti-CD4 monoclonal antibody (clone GK1.5, which blocks class II MHC antigen-specific binding to CD4 and thereby blocks CD4 T cell function (16)) during co-culture returned the IL-17 levels to those of the control T cells (Fig. 4B), indicating that CD4 T cells are the predominant source of IL-17 in this system. We also isolated intraparenchymal and airway lung leukocytes 18 h after challenge of immune and non-immune mice by digesting the lung tissue with collagenase and DNase followed by a discontinuous Percoll gradient. Numbers of T cells in the lungs prior to challenge were too low to allow accurate assessment of cytokine secretion from cells prior to challenge so we therefore only evaluated cytokine secretion from lung T cells after challenge. At the 18-h time point after challenge, the cellular infiltrate is approximately 35% lymphocytes (with the remainder neutrophils) as assessed by forward-and side-scatter FACS analysis; and the composition was not significantly different between the experimental groups (not shown). The isolated lung leukocytes were co-cultured with gentamicin-killed PAO1 for 7 days. The IL-17 levels in the supernatants of these in vivo-immunized, ex vivo-stimulated lung leukocytes are shown in Fig. 4C and were remarkably higher in the ΔaroA-immune groups, with the PAO1ΔaroA-immune and PA14ΔaroA-immune T cells having approximately 10-fold and 40-fold higher IL-17, respectively, than the control T cell groups.
Fig. 4.

Nasal immunization with P. aeruginosa ΔaroA vaccine strains elicits Th17 cells in the spleen and lung. A. IL-17 (upper panel) and IFN-γ (lower panel) production by splenic T cells isolated 3 weeks after nasal immunization and then stimulated for 7 days in the presence of gentamicin-killed whole bacterial cells (MOI: 10 bacteria per T cell) of P. aeruginosa strains PAO1 or PA14 along with irradiated splenocytes. Cells were pooled from 3-5 mice per group. B. IL-17 secretion decreases to baseline when splenic T cells are co-cultured as in (A.) with strain PAO1 along with anti-CD4 monoclonal antibody (clone GK1.5, 1 μg/well). IC, isotype control IgG2b monoclonal antibody (1 μg/well). Bars indicate means of triplicate wells, error bars the SEM. #p<0.05 compared with E. coli control group by ANOVA with Dunnett's multiple comparison test. *p<0.01 compared with corresponding vaccine group treated with IC by ANOVA with Tukey's multiple comparison test. C. IL-17 production by lung leukocytes isolated from vaccinated and unvaccinated mice (3 mice per group)18 h after challenge with strain ExoU+ PAO1 and then co-cultured for 7 days with gentamicin-killed PAO1 whole bacterial cells (MOI: 10 bacteria per T cell). Bars indicate means of triplicate wells, error bars the SEM. #p<0.01 and *p<0.05 compared with E. coli control group by ANOVA with Dunnett's multiple comparison test. D. Intracellular staining for IL-17 in lymphocytes isolated from collagenase-digested lungs of vaccinated and unvaccinated mice 2 h after challenge with P. aeruginosa strain ExoU+ PAO1. Cells were pooled from 3 mice per group.
To determine the cellular source of the IL-17 in vivo, we measured intracellular staining for IL-17 in lung parenchymal and airway lymphocytes isolated as described above but from mice sacrificed just 2 h after challenge with live bacteria. Again, numbers of lymphocytes in the lungs of immunized mice prior to challenge were too low to allow intracellular cytokine analysis. Cells were stimulated for 15 h with PMA and ionomycin in the presence of brefeldin A. As shown in Fig. 4D, there was a small population of IL-17-producing TCRβ-positive cells in the lungs of ΔaroA-immune mice, with 3-fold more in the PA14ΔaroA-immunized group. Similar proportions of IL-17-positive cells were seen in CD3ε-positive cells, and no IL-17-positive cells were seen outside the lymphocyte gate of ΔaroA-immune mice (not shown). We were unable to assess CD4-staining in these experiments due to apparent decreased expression of CD4 during stimulation (not shown). Unstimulated cells showed no detectable IL-17 (not shown). Lung leukocytes (lymphocytes and non-lymphocytes) isolated 18 h after challenge with live bacteria showed no intracellular staining for IL-17 with or without PMA/ionomycin stimulation (not shown).
Neutrophil recruitment to the lungs of P. aeruginosa ΔaroA-immunized mice
Because IL-17 is known to stimulate neutrophil recruitment to the lung, we tested whether the numbers of neutrophils recruited to the airways are greater in the P. aeruginosa ΔaroA-immunized mice and whether the different P. aeruginosa ΔaroA vaccines elicited different neutrophil recruitment. As shown in Fig. 5A, there were significantly more neutrophils in BALF 6 h after challenge of P. aeruginosa ΔaroA-immunized mice compared with the E. coli-immunized and unimmunized controls. Notably, even at this early time point after challenge, there were significantly fewer CFU of P. aeruginosa in the BALF (Fig. 5B) and homogenized lung tissue (not shown) in the P. aeruginosa ΔaroA-immunized mice compared with the control groups. IL-17 levels in BALF (Fig. 5C) obtained 6 h after challenge with a lethal dose of ExoU+ PAO1 were significantly higher in the PA14ΔaroA-immunized compared with the E. coli-immunized mice (p <0.01, ANOVA with Dunnett's multiple comparison test). At the later time point (18 h), neutrophil numbers and IL-17 levels in BALF were highest in the E. coli-immunized mice. Bacterial CFU were very high in the control groups at this time after challenge, indicating that these neutrophils were not effective at clearing the challenge strain.
Fig. 5.
Increased neutrophil recruitment to airways early after bacterial challenge is coincident with lower bacterial CFU and higher IL-17 levels in BALF. Total BALF neutrophils (A) and bacterial CFU (B) isolated after infection of immune and non-immune mice (n=5-10 mice per group) with P. aeruginosa strain ExoU+ PAO1 (5×106 CFU). Bars represent medians and error bars the interquartile range. P values are by Kruskal-Wallis with Dunn's multiple comparison test. C. IL-17 concentration in BALF following infection of immune and non-immune mice (n=5-15 mice per group) with strain ExoU+ PAO1 (5×106 CFU). Bars depict means and error bars the SEM. *p<0.05 compared with E. coli control group by ANOVA with Dunnett's multiple comparison test.
Effect of loss of IL-17 on protective efficacy of P. aeruginosa ΔaroA vaccines against LPS-heterologous strain challenge
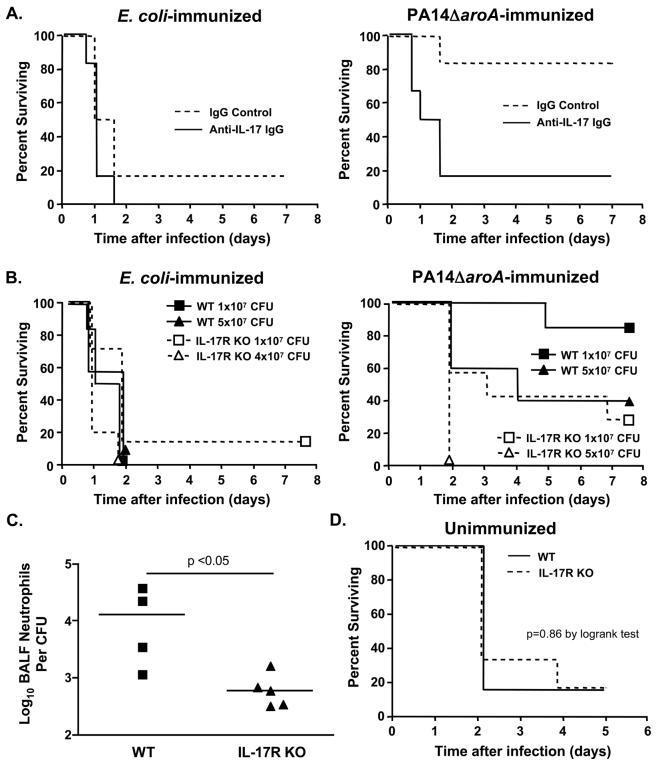
We next determined the effects of neutralization of IL-17 prior to lung challenge with the LPS-heterologous strain PAO1 ExoU+ in mice immunized intranasally with the P. aeruginosa PA14ΔaroA vaccine or with E. coli as control. As expected, there was no effect of neutralization of IL-17 on survival following challenge of the E. coli-immunized mice (left panel of Fig. 6A). However, there was significantly higher mortality (p <0.02, log rank test) in the PA14ΔaroA-immunized mice that received anti-IL-17 IgG compared with the mice given control IgG (Fig. 6A, right panel), indicating that depletion of IL-17 abrogates vaccine-induced protection. In contrast, mice immunized with the PAO1ΔaroA vaccine and then similarly depleted of IL-17 had no change in median survival and were fully protected against LPS-homologous strain PAO1 ExoU+ because these mice had high levels of serum opsonic antibody specific for the LPS of strain PAO1 (not shown). We also found that when we immunized mice genetically deficient in production of the IL-17 receptor (IL-17R KO) with PA14ΔaroA (or with E. coli as control) and then challenged them with ExoU+ PAO1, the IL-17R KO mice had significantly decreased survival compared with wild-type mice (Fig. 6B, right panel). We observed lower percent survival in the PA14ΔaroA-immunized wild-type C57BL/6 mice challenged with 5×107 CFU/mouse (40% survival, Fig. 6B) compared with PA14ΔaroA-immunized wild-type C3H/HeN mice given the same challenge dose (88%, Table 2). This was likely due to the high susceptibility of mice in the C57BL/6 background to P. aeruginosa lung infection (17) and the relative resistance of C3H/HeN mice (18).
Fig. 6.
Neutralization of IL-17 or absence of its receptor diminishes vaccine-induced protection from P. aeruginosa pneumonia. A. Survival of immunized wild-type C3H/HeN mice after administration of IL-17-specific rabbit IgG (solid line) or control IgG (dashed line) 1 day prior to challenge with ExoU+ PAO1 (n=6 mice/group, 5×107 CFU). B. Survival of immunized wild-type C57BL/6 (WT, solid line) or IL-17R KO (dotted line) mice after challenge with ExoU+ PAO1 (n=5-7 mice per group; p<0.05 by logrank test for comparison of PA14ΔaroA WT vs. PA14ΔaroA IL-17R KO mice at either challenge dose). C. Neutrophil recruitment in PA14ΔaroA-immunized mice is deficient in the absence of IL-17R signaling. BALF total neutrophil numbers were measured 6 h after challenge with ExoU+ PAO1 (7×106 CFU) and normalized to bacterial CFU. Each point represents one mouse. P value by Mann Whitney U test. D. Innate immunity to P. aeruginosa pneumonia is intact in IL-17R KO mice. Survival of unimmunized wild-type C57BL/6 (solid line) and IL-17R KO (dashed line) mice after challenge of with ExoU+ PAO1 (n=6 mice per group, 2×106 CFU). All mice (6 per group) survived after a challenge dose of 7×105 CFU (not shown).
Consistent with the IL-17 neutralization experiments in PAO1ΔaroA-immunized C3H/HeN mice, IL-17R KO mice immunized with the PAO1ΔaroA vaccine and then challenged with the LPS-homologous strain ExoU+ PAO1 were fully protected (not shown). These PAO1ΔaroA-immunized IL-17R KO mice had normal levels of opsonic antibody to strain PAO1 (not shown), which very likely accounted for the lack of an impact of deficient IL-17 signaling. We next evaluated the neutrophil recruitment to the airways 6 h after challenge of PA14ΔaroA-immunized wild-type C57BL/6 mice compared to PA14ΔaroA-immunized IL-17R KO mice. We again used P. aeruginosa strain ExoU+ PAO1 (7×106 CFU/mouse) as the challenge strain. As shown in Fig. 6C, there were significantly fewer BALF neutrophils normalized to bacterial load in the IL-17R KO mice. Of note, unimmunized IL-17R KO and wild-type C57BL/6 mice had similar survival curves when challenged with 2×106 CFU of strain ExoU+ PAO1 (Fig. 6D) or 7×105 CFU (100% survival in both groups, n=6 mice per group, not shown), indicating that IL-17R signaling is not important for the innate host defense against acute lethal pneumonia caused by P. aeruginosa.
Discussion
In the current study, we tested the hypothesis that a live-attenuated P. aeruginosa vaccine based on the broad-host-range P. aeruginosa strain PA14 would elicit a broader array of immune effectors than a vaccine derived from strain PAO1. We found that unlike the PAO1ΔaroA vaccine, active immunization with the PA14ΔaroA vaccine could protect against acute lethal pneumonia caused by LPS-heterologous P. aeruginosa strains. Surprisingly, we observed that this protection was not due to serum opsonic activity but rather was dependent on the T cell-secreted cytokine IL-17. The protective efficacy was coincident with a rapid reduction in bacterial CFU in BALF as well as higher neutrophil numbers and higher IL-17 in the BALF 6 h after lung challenge. These findings indicate that active immunization with P. aeruginosa vaccines that induce T cells secreting IL-17 could be a key component of full immunity to lung infection by this pathogen and, furthermore, might be an explanation for why a large clinical trial of passively administered hyperimmune IgG obtained from volunteers given a multivalent P. aeruginosa O-antigen conjugate vaccine did not reduce the incidence of infection in recipients of the antibody (5).
Recent evidence suggests that IL-17-secreting CD4+ T helper cells comprise a distinct lineage called Th17 (19, 20) and play an important role in innate antibacterial host defense in the lung (21-23) as well as in autoimmunity (24). IL-17 is a proinflammatory cytokine expressed primarily by activated memory CD4+ T cells and is the prototypic member of a family of cytokines named IL-17A-F (where IL-17A is what is typically denoted IL-17, and IL-17E is IL-25) (25). IL-17-secreting NKT cells (specifically, CD1d-restricted, NK1.1-negative NKT cells with invariant TCR) have also recently been shown to recognize glycolipid ligands derived from Sphingomonas wittichii and Borrelia burgdorferi and to be involved in neutrophil recruitment to the airways after exposure to LPS (26). Th17 cell development has been linked to IL-23, an IL-12 cytokine family member that shares a common subunit with IL-12 (IL-12p40) but has a distinct subunit, p19 (27). TGF-β appears to be critical for commitment to Th17 development by increasing IL-23R expression, thereby conferring responsiveness to IL-23 (28). An inflammatory milieu (in particular the proinflammatory cytokine IL-6, with amplification by IL-1β and TNF-α) is also required Th17 polarization (29).
The mechanisms whereby the PA14ΔaroA vaccine induces more robust IL-17 responses than the PAO1ΔaroA vaccine after subsequent challenge with live bacteria are not clear. Presumably, the PA14-based vaccine induces higher levels of IL-23, IL-6, IL-1β, TNF-α, and/or TGF-β during the priming of the immune response in the lung or local lymph nodes. This could be related to differential inflammatory activity of the different LPS O antigens of the two vaccine strains and/or to one or several of the additional genes and gene clusters possessed by strain PA14 that are absent from strain PAO1, including the pathogenicity islands PAPI-1 (108 kb) and PAPI-2 (11 kb) (30). Notably, a subtractive hybridization technique called representational difference analysis found not only that several genes, including pilA and pilC, in strain PA14 differed substantially from their counterparts in strain PAO1 but also that only strain PA14 possessed a homolog of the gene for the siderophore-associated ABC transporter protein YbtQ of Yersinia pestis that, when mutated, led to significant attenuation in G. mellonella and in a burned mouse model of sepsis (31). While some siderophores are known to suppress T cell proliferation and function (32) others such as the siderophore lactoferrin have been shown to induce T helper responses to the BCG vaccine (33), so it is conceivable that differences in siderophore production might skew a vaccine-induced T cell response.
The action of IL-17 in the lung centers on neutrophil recruitment via induction of CXC chemokine secretion (KC and MIP-2 in rodents, GRO and IL-8 in humans) along with granulopoietic factors (such as G-CSF) that lead to increased bone marrow production and/or prolonged survival of neutrophils (21). In studies of innate immunity to Klebsiella pneumoniae, Kolls and co-workers found that IL-17R-deficient mice were highly susceptible in an acute pneumonia model, with increased mortality and bacterial dissemination associated with delayed neutrophil recruitment to the lung as well as lower levels of G-CSF and MIP-2 in the knockout mice (23, 34). In acute P. aeruginosa pneumonia, on the other hand, we found that non-immune IL-17R-deficient mice had similar susceptibility compared with wild-type mice, although the IL-17R-deficient mice were much more susceptible in the setting of vaccination. In a different lung infection model, the Kolls group has shown that IL-23-deficient mice had less lung inflammation after challenge with a mucoid P. aeruginosa strain encased in agar beads (35).
Overexpression of IL-17 in the lung via a recombinant adenoviral vector led to local induction of TNF-α, MIP-2, and G-CSF and augmented neutrophil recruitment and bacterial clearance after challenge with K. pneumoniae (22). In this model, signaling through TLR4 was shown to be critical for IL-23 generation (36). Interestingly, CF patients were found to have high levels of IL-17 in bronchoalveolar lavage fluid (BALF), particularly during pulmonary exacerbations (37). Whether IL-17 plays a role in the excessive neutrophilic infiltration and IL-8 secretion characteristic of CF lung disease (38) remains to be determined.
While the time course of IL-17 production in innate immunity models is relatively slow (12-16 h) (23), the production of IL-17 in settings wherein the adaptive response has been activated is far more rapid (as early as 3 h) (39). There is a growing body of evidence that there is a critical role of IL-17 in adaptive immunity to extracellular bacterial respiratory pathogens, particularly Streptococcus pneumoniae and Bordetella pertussis. Malley and co-workers have shown that IL-17 mediates antibody-independent, vaccine-induced protection against nasopharyngeal pneumococcal colonization in mice immunized with the species-common cell wall polysaccharide called the C polysaccharide (40). Higgins et al. have demonstrated that T cell secretion of IL-17 is critical for protective efficacy of the whole-cell B. pertussis vaccine and is also dependent on intact TLR4 signaling (41). Antibody-mediated neutralization of IL-17 in the pertussis studies resulted in an approximately 10-fold increase in CFU 10 days after infection, but the impact on survival was not assessed. Those workers also demonstrated that exposure of peritoneal macrophages or macrophage cell lines to IL-17 enhanced bactericidal activity (41). Evaluation of IL-17R-deficient mice has not been reported in either of these models.
A number of investigators have demonstrated T cell-mediated control of P. aeruginosa infections in rodent models. A high-molecular-weight polysaccharide antigen prepared from the LPS O antigen elicited P. aeruginosa-specific T cells capable of killing the organism in vitro and protecting neutropenic mice against infection in a mechanism whereby anti-LPS IgG on the T cell surface mediated antibody-dependent cellular cytotoxicity (42). Other workers have observed CD4+ T cell-mediated control of P. aeruginosa pneumonia after vaccination with paraformaldehyde-killed P. aeruginosa given to rats orally or injected into Peyer's patches (43, 44). Intravenous immunization of mice with P. aeruginosa-pulsed dendritic cells has also been shown to protect against subsequent P. aeruginosa pneumonia (45). The P. aeruginosa-protective T cell epitopes have not been defined, although studies in humans in the early 1980s reported that the alkaline phosphatase of P. aeruginosa, a secreted protein, was the principle antigen recognized by clones of T cells derived by stimulation of peripheral blood mononuclear cells from healthy volunteers using crude bacterial culture fluids (46). More recently, immunization with a replication-deficient adenoviral vector expressing a 14-amino acid epitope of P. aeruginosa outer membrane protein OprF showed that CD4+ and CD8+ T cell responses to the OprF peptide could be elicited as well as protection against lethal pneumonia in mice (47). Investigations into whether Th1 (IFN-γ dominated) versus Th2 (IL-4 and IL-10 dominated) CD4+ T cell responses are more important for protective immunity against P. aeruginosa lung infections have yielded conflicting results, with some reports suggesting that Th1-prone strains of mice are more susceptible and other reports concluding just the opposite (14, 17, 18, 48). None of those studies evaluated Th17 cells or the cytokine IL-17 as they preceded identification of the Th17 lineage.
The importance of a clear understanding of the role of IL-17 in vaccine-induced protection against bacterial pneumonia relates not only to the design of more effective vaccines but also to the fact that inhibitors of the IL-17 pathway are under active development as anti-inflammatory agents for treatment of a host of autoimmune disorders ranging from rheumatoid arthritis to multiple sclerosis (49, 50). Our results suggest that disabling the IL-17 response could nullify the protective effects of some vaccines – a situation that will need to be studied extensively and weighed carefully in the risk-benefit analysis of IL-17-directed treatment of autoimmune diseases.
The present study suggests that IL-17 is a critical component of vaccine-induced immunity to LPS-heterologous strains of P. aeruginosa and thus paves the way towards a broadly protective P. aeruginosa vaccine using either mixtures of multiple live-attenuated aroA deletants or potentially by expressing heterologous LPS O antigens from a single live-attenuated strain such as PA14ΔaroA. The importance of the neutrophil in control of P. aeruginosa infections is also underscored by our results, as is the critical interplay between components of innate and adaptive immunity (neutrophils with T cells and antibody) during vaccine-induced protection from infection.
Acknowledgments
This work was supported by NIH grants K08 AI50036 and R21 HL079423 to GPP, R01AI068112 to JBG, and AI22535 to GBP.
Footnotes
Disclosures
The authors declare no conflict of interest.
References
- 1.Safdar N, Dezfulian C, Collard HR, Saint S. Clinical and economic consequences of ventilator-associated pneumonia: a systematic review. Crit. Care Med. 2005;33:2184–2193. doi: 10.1097/01.ccm.0000181731.53912.d9. [DOI] [PubMed] [Google Scholar]
- 2.Rello J, Rue M, Jubert P, Muses G, Sonora R, Valles J, Niederman MS. Survival in patients with nosocomial pneumonia: impact of the severity of illness and the etiologic agent. Crit. Care Med. 1997;25:1862–1867. doi: 10.1097/00003246-199711000-00026. [DOI] [PubMed] [Google Scholar]
- 3.Burns JL, Gibson RL, McNamara S, Yim D, Emerson J, Rosenfeld M, Hiatt P, McCoy K, Castile R, Smith AL, Ramsey BW. Longitudinal assessment of Pseudomonas aeruginosa in young children with cystic fibrosis. J. Infect. Dis. 2001;183:444–452. doi: 10.1086/318075. [DOI] [PubMed] [Google Scholar]
- 4.Hatano K, Pier GB. Complex serology and immune response of mice to variant high-molecular-weight O polysaccharides isolated from Pseudomonas aeruginosa serogroup O2 strains. Infect. Immun. 1998;66:3719–3726. doi: 10.1128/iai.66.8.3719-3726.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Donta ST, Peduzzi P, Cross AS, Sadoff J, Haakenson C, Cryz SJ, Jr., Kauffman C, Bradley S, Gafford G, Elliston D, Beam TR, Jr., John JF, Jr., Ribner B, Cantey R, Welsh CH, Ellison RT, 3rd, Young EJ, Hamill RJ, Leaf H, Schein RM, Mulligan M, Johnson C, Abrutyn E, Griffiss JM, Slagle D, T. F. H. I. T. S. Group Immunoprophylaxis against Klebsiella and Pseudomonas aeruginosa infections. The Federal Hyperimmune Immunoglobulin Trial Study Group. J. Infect. Dis. 1996;174:537–543. doi: 10.1093/infdis/174.3.537. [DOI] [PubMed] [Google Scholar]
- 6.Priebe GP, Brinig MM, Hatano K, Grout M, Coleman FT, Pier GB, Goldberg JB. Construction and characterization of a live, attenuated aroA deletion mutant of Pseudomonas aeruginosa as a candidate intranasal vaccine. Infect. Immun. 2002;70:1507–1517. doi: 10.1128/IAI.70.3.1507-1517.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Priebe GP, Meluleni GJ, Coleman FT, Goldberg JB, Pier GB. Protection against fatal Pseudomonas aeruginosa pneumonia in mice after nasal immunization with a live, attenuated aroA deletion mutant. Infect. Immun. 2003;71:1453–1461. doi: 10.1128/IAI.71.3.1453-1461.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mahajan-Miklos S, Rahme LG, Ausubel FM. Elucidating the molecular mechanisms of bacterial virulence using non-mammalian hosts. Mol. Microbiol. 2000;37:981–988. doi: 10.1046/j.1365-2958.2000.02056.x. [DOI] [PubMed] [Google Scholar]
- 9.Chung DR, Kasper DL, Panzo RJ, Chitnis T, Grusby MJ, Sayegh MH, Tzianabos AO. CD4+ T cells mediate abscess formation in intra-abdominal sepsis by an IL-17-dependent mechanism. J. Immunol. 2003;170:1958–1963. doi: 10.4049/jimmunol.170.4.1958. [DOI] [PubMed] [Google Scholar]
- 10.Allewelt M, Coleman FT, Grout M, Priebe GP, Pier GB. Acquisition of expression of the P. aeruginosa ExoU cytotoxin leads to increased bacterial virulence in a murine model of acute pneumonia and systemic spread. Infect. Immun. 2000;68:3998–4004. doi: 10.1128/iai.68.7.3998-4004.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Masopust D, Vezys V, Marzo AL, Lefrancois L. Preferential localization of effector memory cells in nonlymphoid tissue. Science. 2001;291:2413–2417. doi: 10.1126/science.1058867. [DOI] [PubMed] [Google Scholar]
- 12.Priebe GP, Pier GB. Vaccines for Pseudomonas aeruginosa. In: Ellis RW, Brodeur BR, editors. New Bacterial Vaccines. Landes Bioscience; Georgetown, TX: 2003. pp. 261–283. [Google Scholar]
- 13.Priebe GP, Dean CR, Zaidi T, Meluleni GJ, Coleman FT, Coutinho YS, Noto MJ, Urban TA, Pier GB, Goldberg JB. The galU gene of Pseudomonas aeruginosa is required for corneal infection and efficient systemic spread following pneumonia but not for infection confined to the lung. Infect. Immun. 2004;72:4224–4232. doi: 10.1128/IAI.72.7.4224-4232.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jensen PO, Moser C, Kobayashi O, Hougen HP, Kharazmi A, Hoiby N. Faster activation of polymorphonuclear neutrophils in resistant mice during early innate response to Pseudomonas aeruginosa lung infection. Clin. Exp. Immunol. 2004;137:478–485. doi: 10.1111/j.1365-2249.2004.02554.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scarff JM, Goldberg JB. Vaccination against Pseudomonas aeruginosa pneumonia in immunocompromised mice. Clin. Vaccine Immunol. 2007;15:367–375. doi: 10.1128/CVI.00419-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wilde DB, Marrack P, Kappler J, Dialynas DP, Fitch FW. Evidence implicating L3T4 in class II MHC antigen reactivity; monoclonal antibody GK1.5 (anti-L3T4a) blocks class II MHC antigen-specific proliferation, release of lymphokines, and binding by cloned murine helper T lymphocyte lines. J. Immunol. 1983;131:2178–2183. [PubMed] [Google Scholar]
- 17.Stevenson MM, Kondratieva TK, Apt AS, Tam MF, Skamene E. In vitro and in vivo T cell responses in mice during bronchopulmonary infection with mucoid Pseudomonas aeruginosa. Clin. Exp. Immunol. 1995;99:98–105. doi: 10.1111/j.1365-2249.1995.tb03478.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Moser C, Johansen HK, Song Z, Hougen HP, Rygaard J, Hoiby N. Chronic Pseudomonas aeruginosa lung infection is more severe in Th2 responding BALB/c mice compared to Th1 responding C3H/HeN mice. APMIS. 1997;105:838–842. [PubMed] [Google Scholar]
- 19.Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 2005;6:1123–1132. doi: 10.1038/ni1254. [DOI] [PubMed] [Google Scholar]
- 20.Steinman L. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 2007;13:139–145. doi: 10.1038/nm1551. [DOI] [PubMed] [Google Scholar]
- 21.Kolls JK, Kanaly ST, Ramsay AJ. Interleukin-17: an emerging role in lung inflammation. Am. J. Respir. Cell. Mol. Biol. 2003;28:9–11. doi: 10.1165/rcmb.2002-0255PS. [DOI] [PubMed] [Google Scholar]
- 22.Ye P, Garvey PB, Zhang P, Nelson S, Bagby G, Summer WR, Schwarzenberger P, Shellito JE, Kolls JK. Interleukin-17 and lung host defense against Klebsiella pneumoniae infection. Am. J. Respir. Cell. Mol. Biol. 2001;25:335–340. doi: 10.1165/ajrcmb.25.3.4424. [DOI] [PubMed] [Google Scholar]
- 23.Ye P, Rodriguez FH, Kanaly S, Stocking KL, Schurr J, Schwarzenberger P, Oliver P, Huang W, Zhang P, Zhang J, Shellito JE, Bagby GJ, Nelson S, Charrier K, Peschon JJ, Kolls JK. Requirement of interleukin-17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. J. Exp. Med. 2001;194:519–527. doi: 10.1084/jem.194.4.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J. Immunol. 2006;177:566–573. doi: 10.4049/jimmunol.177.1.566. [DOI] [PubMed] [Google Scholar]
- 25.Aggarwal S, Gurney AL. IL-17: prototype member of an emerging cytokine family. J. Leukoc. Biol. 2002;71:1–8. [PubMed] [Google Scholar]
- 26.Michel M-L, Keller1 AC, Paget C, Fujio M, Trottein F, Savage PB, Wong C-H, Schneider E, Dy M, Leite-de-Moraes MC. Identification of an IL-17–producing NK1.1neg iNKT cell population involved in airway neutrophilia. J. Exp. Med. 2007;204:995–1001. doi: 10.1084/jem.20061551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Weaver CT, Hatton RD, Mangan PR, Harrington LE. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu. Rev. Immunol. 2007;25:821–852. doi: 10.1146/annurev.immunol.25.022106.141557. [DOI] [PubMed] [Google Scholar]
- 28.Mangan PR, Harrington LE, O'Quinn DB, Helms WS, Bullard DC, Elson CO, Hatton RD, Wahl SM, Schoeb TR, Weaver CT. Transforming growth factor-beta induces development of the T(H)17 lineage. Nature. 2006;441:231–234. doi: 10.1038/nature04754. [DOI] [PubMed] [Google Scholar]
- 29.Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B. TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity. 2006;24:179–189. doi: 10.1016/j.immuni.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 30.He J, Baldini RL, Deziel E, Saucier M, Zhang Q, Liberati NT, Lee D, Urbach J, Goodman HM, Rahme LG. The broad host range pathogen Pseudomonas aeruginosa strain PA14 carries two pathogenicity islands harboring plant and animal virulence genes. Proc. Nat. Acad. Sci. U.S.A. 2004;101:2530–2535. doi: 10.1073/pnas.0304622101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Choi JY, Sifri CD, Goumnerov BC, Rahme LG, Ausubel FM, Calderwood SB. Identification of virulence genes in a pathogenic strain of Pseudomonas aeruginosa by representational difference analysis. J. Bacteriol. 2002;184:952–961. doi: 10.1128/jb.184.4.952-961.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Autenrieth I, Hantke K, Heesemann J. Immunosuppression of the host and delivery of iron to the pathogen: a possible dual role of siderophores in the pathogenesis of microbial infections? Med. Microbiol. Immunol. 1991;180:135–141. doi: 10.1007/BF00206117. [DOI] [PubMed] [Google Scholar]
- 33.Hwang SA, Kruzel ML, Actor JK. Lactoferrin augments BCG vaccine efficacy to generate T helper response and subsequent protection against challenge with virulent Mycobacterium tuberculosis. Int Immunopharmacol. 2005;5:591–599. doi: 10.1016/j.intimp.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 34.Happel KI, Zheng M, Young E, Quinton LJ, Lockhart E, Ramsay AJ, Shellito JE, Schurr JR, Bagby GJ, Nelson S, Kolls JK. Cutting Edge: Roles of Toll-Like Receptor 4 and IL-23 in IL-17 expression in response to Klebsiella pneumoniae infection. J. Immunol. 2003;170:4432–4436. doi: 10.4049/jimmunol.170.9.4432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dubin PJ, Kolls JK. IL-23 mediates inflammatory responses to mucoid Pseudomonas aeruginosa lung infection in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2007;292:L519–528. doi: 10.1152/ajplung.00312.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Happel KI, Lockhart EA, Mason CM, Porretta E, Keoshkerian E, Odden AR, Nelson S, Ramsay AJ. Pulmonary interleukin-23 gene delivery increases local T-cell immunity and controls growth of Mycobacterium tuberculosis in the lungs. Infect. Immun. 2005;73:5782–5788. doi: 10.1128/IAI.73.9.5782-5788.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McAllister F, Henry A, Kreindler JL, Dubin PJ, Ulrich L, Steele C, Finder JD, Pilewski JM, Carreno BM, Goldman SJ, Pirhonen J, Kolls JK. Role of IL-17A, IL-17F, and the IL-17 receptor in regulating growth-related oncogene-alpha and granulocyte colony-stimulating factor in bronchial epithelium: implications for airway inflammation in cystic fibrosis. J. Immunol. 2005;175:404–412. doi: 10.4049/jimmunol.175.1.404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.DiMango E, Ratner AJ, Bryan R, Tabibi S, Prince A. Activation of NF-kappaB by adherent Pseudomonas aeruginosa in normal and cystic fibrosis respiratory epithelial cells. J. Clin. Invest. 1998;101:2598–2605. doi: 10.1172/JCI2865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hellings PW, Kasran A, Liu Z, Vandekerckhove P, Wuyts A, Overbergh L, Mathieu C, Ceuppens JL. Interleukin-17 orchestrates the granulocyte influx into airways after allergen inhalation in a mouse model of allergic asthma. Am. J. Respir. Cell. Mol. Biol. 2003;28:42–50. doi: 10.1165/rcmb.4832. [DOI] [PubMed] [Google Scholar]
- 40.Malley R, Srivastava A, Lipsitch M, Thompson CM, Watkins C, Tzianabos A, Anderson PW. Antibody-independent, interleukin-17A-mediated, cross-serotype immunity to pneumococci in mice immunized intranasally with the cell wall polysaccharide. Infect. Immun. 2006;74:2187–2195. doi: 10.1128/IAI.74.4.2187-2195.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Higgins SC, Jarnicki AG, Lavelle EC, Mills KH. TLR4 mediates vaccine-induced protective cellular immunity to Bordetella pertussis: role of IL-17-producing T cells. J. Immunol. 2006;177:7980–7989. doi: 10.4049/jimmunol.177.11.7980. [DOI] [PubMed] [Google Scholar]
- 42.Markham RB, Pier GB, Schreiber JR. The role of cytophilic IgG3 antibody in T cell-mediated resistance to infection with the extracellular bacterium, Pseudomonas aeruginosa. J. Immunol. 1991;146:316–320. [PubMed] [Google Scholar]
- 43.Dunkley ML, Clancy RL, Cripps AW. A role for CD4+ T cells from orally immunized rats in enhanced clearance of Pseudomonas aeruginosa from the lung. Immunology. 1994;83:362–369. [PMC free article] [PubMed] [Google Scholar]
- 44.Dunkley ML, Cripps AW, Clancy RL. Immunity to respiratory Pseudomonas aeruginosa infection: P. aeruginosa-specific T cells arising after intestinal immunization. Adv. Exp. Med. Biol. 1995;371B:755–759. [PubMed] [Google Scholar]
- 45.Worgall S, Kikuchi T, Singh R, Martushova K, Lande L, Crystal RG. Protection against pulmonary infection with Pseudomonas aeruginosa following immunization with P. aeruginosa-pulsed dendritic cells. Infect. Immun. 2001;69:4521–4527. doi: 10.1128/IAI.69.7.4521-4527.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Parmely MJ, Iglewski BH, Horvat RT. Identification of the principal T lymphocyte-stimulating antigens of Pseudomonas aeruginosa. J. Exp. Med. 1984;160:1338–1349. doi: 10.1084/jem.160.5.1338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Worgall S, Krause A, Rivara M, Hee K-K, Vintayen EV, Hackett NR, Roelvink PW, Bruder JT, Wickham TJ, Kovesdi I, Crystal RG. Protection against P. aeruginosa with an adenovirus vector containing an OprF epitope in the capsid. J. Clin. Invest. 2005;115:1281–1289. doi: 10.1172/JCI23135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kondratieva TK, Kobets NV, Khaidukov SV, Yeremeev VV, Lyadova IV, Apt AS, Tam MF, Stevenson MM. Characterization of T cell clones derived from lymph nodes and lungs of Pseudomonas aeruginosa-susceptible and resistant mice following immunization with heat-killed bacteria. Clin. Exp. Immunol. 2000;121:275–282. doi: 10.1046/j.1365-2249.2000.01222.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lubberts E, Koenders MI, Oppers-Walgreen B, van den Bersselaar L, Coenen-de Roo CJ, Joosten LA, van den Berg WB. Treatment with a neutralizing anti-murine interleukin-17 antibody after the onset of collagen-induced arthritis reduces joint inflammation, cartilage destruction, and bone erosion. Arthritis Rheum. 2004;50:650–659. doi: 10.1002/art.20001. [DOI] [PubMed] [Google Scholar]
- 50.Rohn TA, Jennings GT, Hernandez M, Grest P, Beck M, Zou Y, Kopf M, Bachmann MF. Vaccination against IL-17 suppresses autoimmune arthritis and encephalomyelitis. Eur J Immunol. 2006;36:2857–2867. doi: 10.1002/eji.200636658. [DOI] [PubMed] [Google Scholar]
- 51.Dean CR, Goldberg JB. Pseudomonas aeruginosa galU is required for a complete lipopolysaccharide core and repairs a secondary mutation in a PA103 (serogroup O11) wbpM mutant. FEMS Microbiol. Lett. 2002;210:277–283. doi: 10.1111/j.1574-6968.2002.tb11193.x. [DOI] [PubMed] [Google Scholar]
- 52.Preston MJ, Fleiszig SM, Zaidi TS, Goldberg JB, Shortridge VD, Vasil ML, Pier GB. Rapid and sensitive method for evaluating Pseudomonas aeruginosa virulence factors during corneal infections in mice. Infect. Immun. 1995;63:3497–3501. doi: 10.1128/iai.63.9.3497-3501.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fleiszig SMJ, Zaidi TS, Preston MJ, Goldberg JB, Grout M, Evans DJ, Pier GB. Relationship between cytotoxicity and corneal epithelial cell invasion by clinical isolates of Pseudomonas aeruginosa. Infect. Immun. 1996;64:2288–2294. doi: 10.1128/iai.64.6.2288-2294.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Boyer HW, Roulland-Dussoix D. A complementation analysis of the restriction and modification of DNA in Escherichia coli. J. Mol. Biol. 1969;41:459–472. doi: 10.1016/0022-2836(69)90288-5. [DOI] [PubMed] [Google Scholar]
- 55.Schweizer HP. Improved broad-host-range lac-based plasmid vectors for the isolation and characterization of protein fusions in Pseudomonas aeruginosa. Gene. 1991;103:87–92. doi: 10.1016/0378-1119(91)90396-s. [DOI] [PubMed] [Google Scholar]
- 56.Schweizer HP. Escherichia-Pseudomonas shuttle vectors derived from pUC18/19. Gene. 1991;97:109–121. doi: 10.1016/0378-1119(91)90016-5. [DOI] [PubMed] [Google Scholar]