Abstract
Purified preparations of 10-nm neurofilaments from rat spinal cord and bovine or porcine brain contain a predominant 68,000-dalton polypeptide. This polypeptide is also a major component of the neurofilaments that copurify with brain tubulin isolated by cycles of polymerization and depolymerization. A protein that has the same isoelectric point and molecular weight as the neurofilament-associated polypeptide has also been identified as a cytoskeletal protein in a variety of avian and mammalian cell types, including baby hamster kidney (BHK-21) mouse 3T3, Novikoff rat hepatoma, chicken fibroblast, and chicken muscle cells. This protein is also a component of isolated chicken skeletal myofibrils. One-dimensional peptide maps of the 68,000-dalton proteins purified by two-dimensional isoelectric focusing/NaDodSO4/polyacrylamide gel electrophoresis from myofibrils, cycled tubulin, purified neurofilaments, and various cultured cell types were identical. In immunofluorescence this protein was associated with cytoplasmic intermediate filaments and myofibril Z discs. These results indicate that the neurofilament-associated polypeptide is a conserved protein that is present in many different cell types in addition to neuronal cells. Because some of these cells contain the major components of two other intermediate filament classes, desmin and vimentin, a given cell type may contain the subunits of at least three distinct intermediate filament types.
Keywords: intermediate filaments, immunofluorescence, two-dimensional isoelectric focusing, sodium dodecyl sulfate, polyacrylamide gel electrophoresis
Full text
PDF


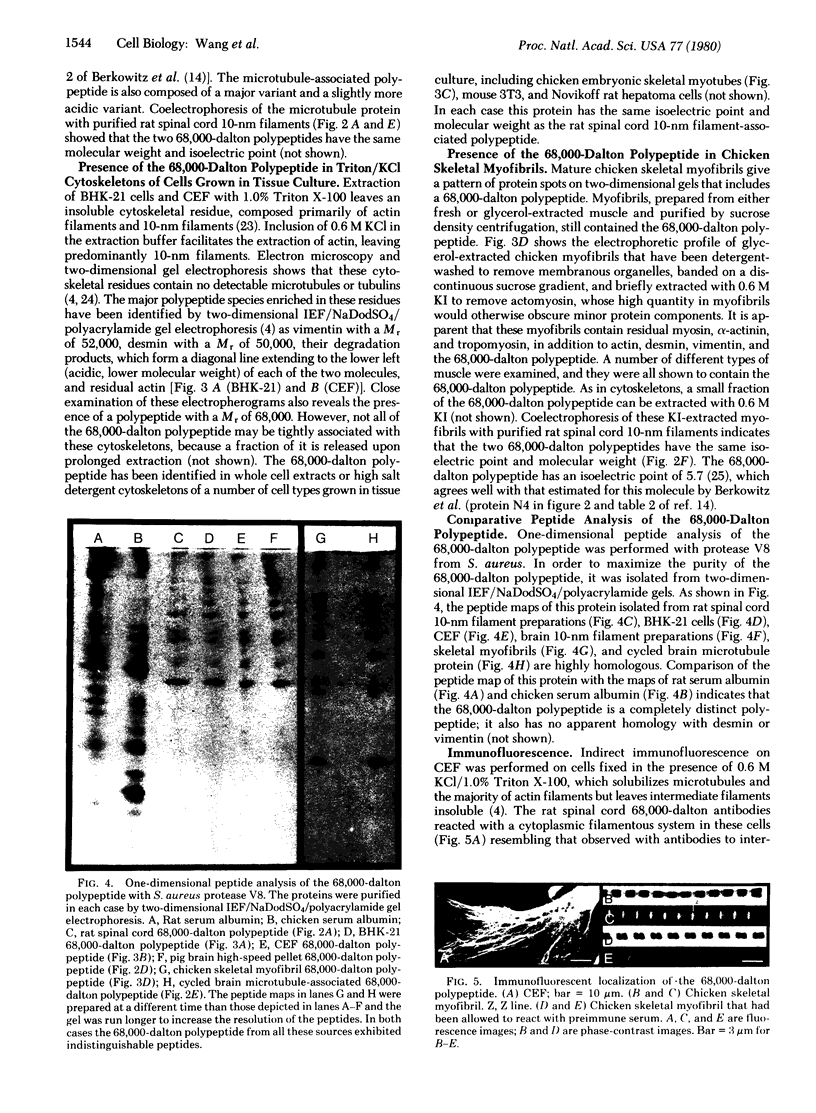

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berkowitz S. A., Katagiri J., Binder H. K., Williams R. C., Jr Separation and characterization of microtubule proteins from calf brain. Biochemistry. 1977 Dec 13;16(25):5610–5617. doi: 10.1021/bi00644a035. [DOI] [PubMed] [Google Scholar]
- Borisy G. G., Olmsted J. B. Nucleated assembly of microtubules in porcine brain extracts. Science. 1972 Sep 29;177(4055):1196–1197. doi: 10.1126/science.177.4055.1196. [DOI] [PubMed] [Google Scholar]
- Brown S., Levinson W., Spudich J. A. Cytoskeletal elements of chick embryo fibroblasts revealed by detergent extraction. J Supramol Struct. 1976;5(2):119–130. doi: 10.1002/jss.400050203. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Franke W. W., Schmid E., Osborn M., Weber K. Different intermediate-sized filaments distinguished by immunofluorescence microscopy. Proc Natl Acad Sci U S A. 1978 Oct;75(10):5034–5038. doi: 10.1073/pnas.75.10.5034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franke W. W., Schmid E., Weber K., Osborn M. HeLa cells contain intermediate-sized filaments of the prekeratin type. Exp Cell Res. 1979 Jan;118(1):95–109. doi: 10.1016/0014-4827(79)90587-1. [DOI] [PubMed] [Google Scholar]
- Gard D. L., Bell P. B., Lazarides E. Coexistence of desmin and the fibroblastic intermediate filament subunit in muscle and nonmuscle cells: identification and comparative peptide analysis. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3894–3898. doi: 10.1073/pnas.76.8.3894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gard D. L., Lazarides E. The synthesis and distribution of desmin and vimentin during myogenesis in vitro. Cell. 1980 Jan;19(1):263–275. doi: 10.1016/0092-8674(80)90408-0. [DOI] [PubMed] [Google Scholar]
- Granger B. L., Lazarides E. Desmin and vimentin coexist at the periphery of the myofibril Z disc. Cell. 1979 Dec;18(4):1053–1063. doi: 10.1016/0092-8674(79)90218-6. [DOI] [PubMed] [Google Scholar]
- Hoffman P. N., Lasek R. J. The slow component of axonal transport. Identification of major structural polypeptides of the axon and their generality among mammalian neurons. J Cell Biol. 1975 Aug;66(2):351–366. doi: 10.1083/jcb.66.2.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubbard B. D., Lazarides E. Copurification of actin and desmin from chicken smooth muscle and their copolymerization in vitro to intermediate filaments. J Cell Biol. 1979 Jan;80(1):166–182. doi: 10.1083/jcb.80.1.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynes R. O., Destree A. T. 10 nm filaments in normal and transformed cells. Cell. 1978 Jan;13(1):151–163. doi: 10.1016/0092-8674(78)90146-0. [DOI] [PubMed] [Google Scholar]
- Lazarides E., Balzer D. R., Jr Specificity of desmin to avian and mammalian muscle cells. Cell. 1978 Jun;14(2):429–438. doi: 10.1016/0092-8674(78)90128-9. [DOI] [PubMed] [Google Scholar]
- Lazarides E. Intermediate filaments as mechanical integrators of cellular space. Nature. 1980 Jan 17;283(5744):249–256. doi: 10.1038/283249a0. [DOI] [PubMed] [Google Scholar]
- Liem R. K., Yen S. H., Salomon G. D., Shelanski M. L. Intermediate filaments in nervous tissues. J Cell Biol. 1978 Dec;79(3):637–645. doi: 10.1083/jcb.79.3.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockwood A. H. Tubulin assembly protein: immunochemical and immunofluorescent studies on its function and distribution in microtubules and cultured cells. Cell. 1978 Apr;13(4):613–627. doi: 10.1016/0092-8674(78)90212-x. [DOI] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Runge M. S., Detrich H. W., 3rd, Williams R. C., Jr Identification of the major 68,000-dalton protein of microtubule preparations as a 10-nm filament protein and its effects on microtubule assembly in vitro. Biochemistry. 1979 May 1;18(9):1689–1698. doi: 10.1021/bi00576a009. [DOI] [PubMed] [Google Scholar]
- Schlaepfer W. W., Freeman L. A. Neurofilament proteins of rat peripheral nerve and spinal cord. J Cell Biol. 1978 Sep;78(3):653–662. doi: 10.1083/jcb.78.3.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer W. W. Immunological and ultrastructural studies of neurofilaments isolated from rat peripheral nerve. J Cell Biol. 1977 Jul;74(1):226–240. doi: 10.1083/jcb.74.1.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer W. W., Micko S. Chemical and structural changes of neurofilaments in transected rat sciatic nerve. J Cell Biol. 1978 Aug;78(2):369–378. doi: 10.1083/jcb.78.2.369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer W. W. Observations on the disassembly of isolated mammalian neurofilaments. J Cell Biol. 1978 Jan;76(1):50–56. doi: 10.1083/jcb.76.1.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer W. W. Studies on the isolation and substructure of mammalian neurofilaments. J Ultrastruct Res. 1977 Nov;61(2):149–157. doi: 10.1016/s0022-5320(77)80081-6. [DOI] [PubMed] [Google Scholar]
- Shelanski M. L., Gaskin F., Cantor C. R. Microtubule assembly in the absence of added nucleotides. Proc Natl Acad Sci U S A. 1973 Mar;70(3):765–768. doi: 10.1073/pnas.70.3.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Starger J. M., Brown W. E., Goldman A. E., Goldman R. D. Biochemical and immunological analysis of rapidly purified 10-nm filaments from baby hamster kidney (BHK-21) cells. J Cell Biol. 1978 Jul;78(1):93–109. doi: 10.1083/jcb.78.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorpe R., Delacourte A., Ayers M., Bullock C., Anderton B. H. The polypeptides of isolated brain 10nm filaments and their association with polymerized tubulin. Biochem J. 1979 Aug 1;181(2):275–284. doi: 10.1042/bj1810275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuszynski G. P., Frank E. D., Damsky C. H., Buck C. A., Warren L. The detection of smooth muscle desmin-like protein in BHK21/C13 fibroblasts. J Biol Chem. 1979 Jul 10;254(13):6138–6143. [PubMed] [Google Scholar]