Abstract
Noroviruses are genetically diverse, uncultivable, positive-sense RNA viruses and are the most common cause of epidemic acute gastroenteritis in humans in the United States. Recent studies of norovirus attachment in vitro by using recombinant virus-like particles (VLPs) suggest that various norovirus strains exhibit different patterns of attachment to ABH histo-blood group antigens, which are carbohydrate epitopes present in high concentrations on mucosal cell surfaces of the gut. However, attachment of live norovirus strains to histo-blood group antigens has not been investigated to date. Utilizing a newly designed magnetic bead-virus capture method, we characterized histo-blood group antigen attachment properties of various norovirus strains obtained from clinical stool specimens to compare the attachment properties of wild-type virus and VLPs and to further map norovirus attachment. Consistent with previous reports using VLPs, various strains of noroviruses exhibited different patterns of attachment to histo- blood group antigens. Norwalk virus bound specifically to H type 1, H type 3, and Leb. Two genogroup II noroviruses, one representing the Toronto genotype and the other from a novel genotype, bound specifically to Leb. A Desert Shield-like strain did not attach to H types 1, 2, or 3, H type 1 and 3 precursors, Lea, or Leb. Surprisingly, wild-type Snow Mountain virus (SMV) attached specifically to H type 3, which contradicted previous findings with SMV VLPs. On further investigation, we found that stool components promote this attachment, providing the first known observation that one or more components of human feces could promote and enhance norovirus attachment to histo-blood group antigens.
Noroviruses are a group of genetically diverse RNA viruses in the family Caliciviridae that account for >90% of outbreaks of acute nonbacterial gastroenteritis in humans in the United States (18, 22). Noroviruses cause an estimated 23 million infections every year nationwide, and although serious complications are not common, infection can result in hospitalization and/or death (40, 43). Noroviruses are transmitted via ingestion of feces-contaminated water and food, exposure to aerosolized feces and vomitus, exposure to contaminated surfaces, and direct person- to-person contact, with disease symptoms including nausea, vomiting, and diarrhea. Outbreaks typically occur in areas of close human contact such as schools, day care centers, medical care facilities, cruise ships, and the military (2, 10, 22).
Because a tissue culture model for human norovirus infection is not available, recombinant molecular approaches have been necessary to dissect their basic biological properties. The genome of Norwalk virus (NV), the prototype norovirus strain, has been cloned, sequenced, and characterized biochemically (35). The genome is a single-stranded, positive- sense RNA of about 7.5 kb and is organized into three open reading frames (ORFs). ORF1 encodes the nonstructural proteins, which include putative polymerase, protease, and helicase motifs. ORF2 encodes a major capsid protein, and ORF3 encodes a minor structural protein (21, 34).
Noroviruses are extremely genetically and antigenically diverse. Based on sequence homologies in the polymerase and capsid genes, the major human strains can be genetically divided into two main genogroups and at least 15 different genotypes (51). In addition, at least three other norovirus genogroups have been identified, which include strains detected in calves (genogroup III), humans (genogroup IV), and mice (genogroup V) (1, 17, 37, 38, 48). The major capsid proteins differ by as much as 30% in amino acid sequence between isolates within a given genogroup and by more than 50% between isolates in different genogroups (23).
The ORF2-encoded major capsid protein self-assembles into virus-like particles (VLPs) when expressed in high concentrations from recombinant baculoviruses in insect cells (34) or from Venezuelan equine encephalitis virus replicon vectors in mammalian cells (5, 27, 29). Norovirus VLPs are morphologically and antigenically indistinguishable from wild-type virus and have provided the foundation for studies of assembly, attachment, and immunobiology. Moreover, they have been indispensable for the development of diagnostic immunoassays and are also the basis for multiple candidate vaccine strategies (4, 5, 25, 27, 29, 36).
Despite the lack of a tissue culture system or an animal model that accurately depicts human norovirus disease, significant advances in norovirus attachment and pathogenesis research have been achieved, including the identification of particular ABH histo-blood group antigens as putative ligands for the attachment of noroviruses to mucosal cell surfaces (27, 33, 42; reviewed in reference 28). These carbohydrate epitopes are present in mucosal secretions and on many tissues throughout the human body. The complexity of the ABH histo-blood group antigens is reflected in the large number of type chain branches and modifying enzymes, as well as the broad distribution of different antigens based on blood type, genetic polymorphisms, and tissue specificity (41). At least five type chains of the ABH histo-blood group antigens have been identified, which are produced and distributed in various tissue compartments in a type- specific manner (47). The type 1 and 3 chain molecules are the predominant ABH histo- blood group antigens on cell surfaces of the gut, which are presumed to be the site(s) of norovirus infection. The FUT2 enzyme, an α1,2-fucosyltransferase, is responsible for the production of type 1 and 3 chain ABH histo-blood group antigens. Approximately 20% of Europeans and Africans express a defective form of the FUT2 enzyme and therefore do not produce type 1 or 3 chain histo-blood group antigens on cell surfaces of the gut or in mucosal secretions; this is known as the “nonsecretor” or secretor-negative phenotype.
The type 1 and 3 H antigens are produced by the FUT2 enzyme in secretor-positive individuals from H precursor molecules. The A or B enzymes produced in blood type A or B individuals, respectively (or both in blood type AB individuals), further modify the H antigens along the particular type chain to produce type-specific A or B antigens. Similarly, the type 2 chain A or B antigens are produced on red blood cells, and the presence or absence of one or more of these molecules is used to determine an individual's blood type. In the type 1 chain, the FUT3 Lewis enzyme, an α1,4-fucosyltransferase, modifies the H type 1 precursor to produce Lea or modifies H type 1 to produce Leb. Approximately 10% of Europeans lack a functional FUT3 gene product, representative of the Lewis-negative phenotype, and therefore will not produce Lea or Leb on mucosal surfaces (41). Secretor-positive, Lewis-positive individuals will produce Leb and relatively less Lea on cell surfaces of the gut and in mucosal secretions, whereas secretor-negative, Lewis-positive individuals will produce only Lea.
Recombinant NV VLPs were first shown to attach to H types 1 and 3 epitopes on the surfaces of gastroduodenal epithelial cells from secretor-positive individuals (42). In addition, we demonstrated that Norwalk VLPs attach specifically to H type 1, H type 3, and Leb carbohydrates, even in the absence of other cellular components (27). The significance of these in vitro findings was recently demonstrated in vivo by Lindesmith et al. using a human challenge model for Norwalk virus infection (39). This study demonstrated that secretor-negative human volunteers, who do not produce H type 1, H type 3, and/or Leb on mucosal cell surfaces, were resistant to live NV challenge. Taken together, the results of these studies suggest that H type 1, H type 3, and Leb are attachment molecules necessary for NV infectivity in the gut.
We have also observed that VLPs of other norovirus strains exhibit different properties of attachment to ABH histo-blood group antigens in vitro (27), suggesting that different norovirus genotypes may utilize different mechanisms for attachment and entry in vivo and that various other host factors may play a role in susceptibility to different norovirus strains. Because VLP reagents are not available for many norovirus strains, the overall extent to which noroviruses utilize ABH histo-blood group antigens for attachment is unclear and cannot be thoroughly evaluated using previously described biochemical methods. Also, no study to date has examined and compared the ability of wild-type virus and recombinant VLPs to attach to specific ABH histo-blood group antigens.
In this study, we developed a method to purify noroviruses from clinical specimens and at the same time characterize their histo-blood group antigen attachment properties. Consistent with our previous observations, various norovirus strains exhibited distinct histo-blood group antigen binding patterns. In addition, we observed that attachment of one particular norovirus strain to H type 3 occurs only in the presence of stool contents, suggesting a potential mechanism of norovirus attachment enhancement by a component of the extracellular environment. The methods and results described in this study may ultimately be useful in mapping the ABH histo-blood group antigen attachment specificities of all Norovirus strains, resulting in the establishment of novel hypotheses regarding norovirus pathogenesis and host susceptibility.
MATERIALS AND METHODS
Norovirus specimens.
Norovirus clinical stool specimens were obtained from individuals challenged with live NV or Snow Mountain virus (SMV) or from various norovirus outbreak samples. The norovirus outbreaks occurred over at least a 5-year period, and the viruses were archived and previously demonstrated to represent various norovirus genotypes (52; C. Moe, unpublished data; J. Vinjé, unpublished data). Various strains from the outbreak specimens were identified and retyped in a blind fashion by reverse transcription-PCR (RT-PCR) followed by DNA sequencing of the RT-PCR products. When necessary to produce a liquid suspension, ∼20% stool/phosphate-buffered saline (PBS) sample solutions were made and clarified by low-speed centrifugation. Viral RNA was extracted from stool samples using Qiamp viral RNA minikits (Qiagen, Valencia, Calif.). A broadly reactive RT-PCR amplification targeting a 327-bp region of the polymerase gene was performed using a One-Step RT-PCR kit (Qiagen) and modified JV12/JV13 primers (52). RT-PCR-positive products were sequenced and aligned to previously published sequences to identify specimens that represent distinct norovirus genotypes. Human challenge specimens and outbreak samples representing different norovirus genotypes were then subjected to titer determination by end-point dilution RT-PCR using the same primers described above, except for Norwalk virus, where strain-specific primers were used (15).
Viral capture and purification method.
A total of 50 μl of streptavidin-coated magnetic bead suspensions (M270; Dynal Biotech, Oslo, Norway) were rinsed three times with 1 ml of sterile PBS in a 1.5-ml microcentrifuge tube. The beads were then preblocked with 5% milk in PBS (5% BLOTTO) for 1 h at ambient temperature on an end-over-end mixer. Synthetic, biotinylated histo-blood group antigens were obtained from Glycotech (Rockville, Md.) and resuspended in PBS at 1 mg/ml. A 50-μl volume of the synthetic carbohydrates was added to the preblocked magnetic beads, and the mixture was incubated at ambient temperature for 2 h on an end-over-end mixer. The beads were then rinsed three times with PBS-Tween (0.05% Tween) and blocked for 1 h at ambient temperature and then overnight at 4°C in 5% BLOTTO containing 0.25% Tween, while continually kept on an end-over-end mixer. The beads were then rinsed three times with PBS-Tween and resuspended in 5% BLOTTO. A norovirus-containing stool specimen (1 to 10 μl) with 105 PCR-detectable units of virus was then added to the bead suspensions, and the mixture was incubated at ambient temperature for 4 h on an end-over-end mixer. The beads were then washed five times with PBS-Tween and three times with sterile PBS and subsequently resuspended in 50 μl of sterile, RNase-free PBS. The washed beads were then 10-fold serially diluted three times and stored at −80°C until tested by RT-PCR.
RT-PCR detection of norovirus attachment.
RT-PCR detection of viral attachment was conducted in a blind fashion, with no prior knowledge of the norovirus strains or histo- blood group carbohydrates being tested for attachment. First, encapsidated viral RNA was heat released from the bead suspensions by incubating 10 μl of each bead dilution at 99°C for 5 min. Then 2.5 μl of the heat-released RNA, as well as RNA extracted and purified from the original stool sample as a positive control, was subjected to RT-PCR using the same primers and conditions for determining the titers of the samples. RT-PCR products were examined on an ethidium bromide-stained 2% agarose gel.
VLP attachment assays.
Two microwell-based assays were designed to detect and quantify norovirus VLP attachment to histo-blood group antigens. In the first assay, synthetic biotinylated carbohydrates (2.5 μg/ml in Tris-buffered saline, 100 μl per well) were added to streptavidin-precoated microwells (Thermo Labsystems, Basingstoke, United Kingdom) and the mixtures were incubated for 2 h at 37°C. The microwells were then blocked with 300 μl of 5% BLOTTO overnight at 4°C. Sucrose gradient-purified norovirus VLPs (5, 27) were then added to the carbohydrate-coated microwells at various concentrations and in the presence or absence of various stool extracts (as indicated in Results). The wells were incubated at 37°C for 4 h and then washed five times with PBS-Tween. To detect attachment, 100 μl of pooled polyclonal antisera from SMV, NV, and Hawaii virus (HV)-infected human volunteers was added per well at a 1:100 dilution in 5% BLOTTO and the wells were incubated at 37°C for 2h. The plates were then washed five times with PBS-Tween, a goat anti-human immunoglobulin G (IgG)-alkaline phosphatase conjugate (1:1,000 in 5% BLOTTO, at 100 μl/well) (Sigma, St. Louis, Mo.) was added, and the mixture was incubated for 1 h at 37°C. After a final washing step, p-nitrophenyl phosphatase substrate (pNPP) (Sigma-FAST tablets; Sigma) was added at 150 μl/well and the plates were incubated for 25 to 30 min at ambient temperature. Developed plates were then read at an optical density of 405 nm (OD405) using a microplate reader (Molecular Devices, Sunnyvale, Calif.). Wells with no VLPs added were used as plate blanks.
A second VLP attachment assay was used to detect the attachment of carbohydrates to VLP-coated microwells, similar to previously described methods (27). Briefly, 2.5 μg of the appropriate VLP reagent per ml (100 μl/well in Tris-buffered saline) was added to high-binding microwells (Costar, Corning, N.Y.) and the plates were incubated for 4 h at ambient temperature and then blocked with 5% BLOTTO overnight at 4°C. Synthetic biotinylated histo-blood group antigens were then added at various concentrations in the presence or absence of stool extracts (as indicated in Results) in 5% BLOTTO, and the plates were incubated at 37°C for 4 h. The plates were then washed five times with PBS-Tween, streptavidin-alkaline phosphatase conjugate (1:500 dilution in 5% BLOTTO, at 100 μl/well) (Sigma) was added, and the mixture was incubated at 37°C for 1 h. Finally, the plates were washed and developed with pNPP and the OD405 was determined. Wells with no carbohydrate added were used as plate blanks. Student's t test (unpaired, two-tailed, unequal variance) was used to determine the statistical significance of stool extract-enhanced attachment of SMV VLPs.
RESULTS
Identification and genotyping of clinical norovirus specimens.
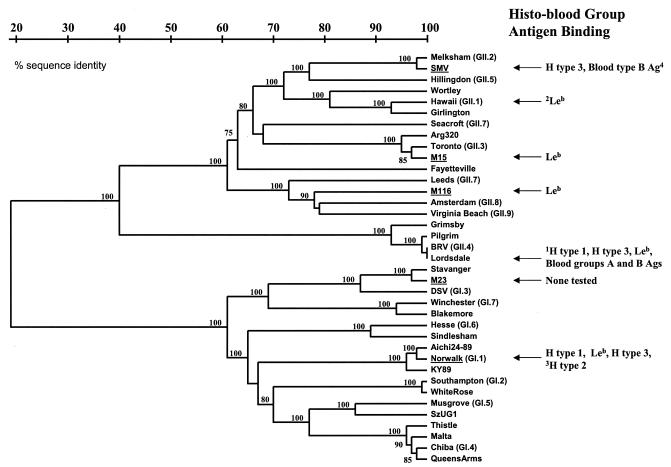
To identify high-titer clinical specimens to be used for this study, we first screened and determined the titers of norovirus stool specimens obtained from human challenge studies or outbreak samples for the presence of noroviruses by RT-PCR using both broadly reactive and NV-specific primer pairs. We then sequenced RT-PCR products from norovirus-positive samples and aligned them with previously published norovirus sequences to determine the genotype of the viruses. Finally, we determined the titers of norovirus stool specimens representing various genotypes by end-point RT-PCR using the same primer sets used to identify the norovirus strains. Stool samples with norovirus titers of 107 to 108 RT-PCR-detectable units were identified and used for attachment characterization assays. For a genetic classification of the strains used in this study and a general summary of attachment data, see Fig. 10.
FIG. 10.
Summary of norovirus attachment data in relation to genetic grouping. Genetic classifications of noroviruses used in this study (indicated by underlining) are based on previously described criteria and compared with known sequences of other noroviruses (genogroup.genotype indicated) (23, 24, 51). The nucleotide sequences that were compared are from a 177-nucleotide region in the ORF2 gene (region D) that has been shown to reliably classify Norovirus genotypes (52). Specific histo-blood group antigen attachment data from this study compiled with results from other studies are indicated. 1Unpublished data obtained using VLP reagents. 2Previous results using HV VLPs alone showed no binding to any carbohydrates tested (27), whereas results from this study demonstrated slight binding of VLPs to Leb, but only in the presence of stool extract. 3Observation made by Hutson et al. using baculovirus-expressed recombinant NV VLPs (33). 4Based on the observation that SMV VLPs attached specifically to saliva from secretor-positive, blood type B individuals (27).
Norovirus capture and histo-blood group antigen attachment characterization.
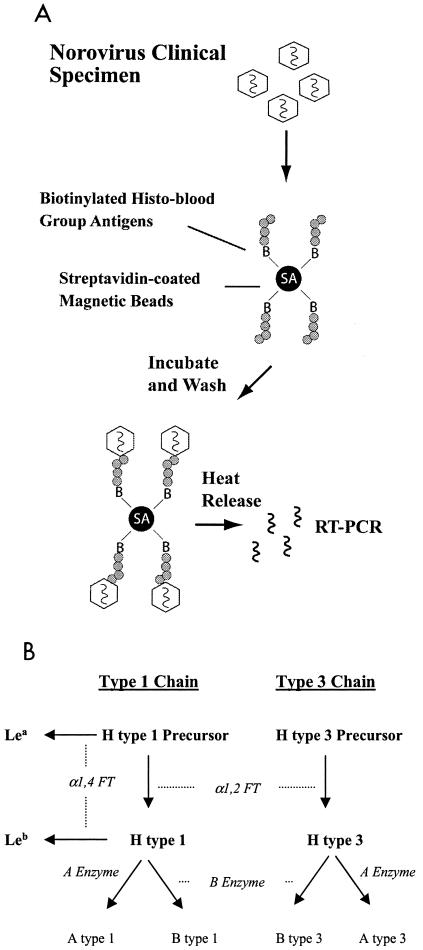
We designed and optimized a magnetic bead-based method to capture and characterize the histo- blood group antigen attachment properties of noroviruses in clinical stool specimens (Fig. 1A). Using this assay, we evaluated the ability of noroviruses representing five different genotypes to attach to synthetic carbohydrates representative of the various histo-blood group antigens that would be present on gut mucosal cell surfaces of individuals of different secretor and Lewis phenotypes (Fig. 1B). The histo-blood group antigen attachment properties of NV, a genogroup I (GI) strain and the prototype norovirus (GI.1 genotype) have been previously characterized using VLP reagents and a variety of biochemical and histological methods (27, 33, 42). Consistent with these studies, NV obtained from a human challenge stool specimen (39) bound specifically to H type 1, Leb, and, to a relatively lesser extent, H type 3 (Fig. 2), suggesting that NV VLPs and wild-type virus are biologically indistinguishable in the case of attachment to these histo-blood group antigens.
FIG. 1.
Magnetic bead method and carbohydrates used in this study. (A) A magnetic bead-based method was designed to characterize histo-blood group antigen attachment specificities of wild-type noroviruses obtained from clinical stool specimens. Streptavidin-precoated magnetic beads were coated with specific synthetic, biotinylated histo-blood group antigens and then blocked with 5% milk-PBS containing 0.25% Tween 20 to inhibit nonspecific binding. Stool extracts containing equal RT-PCR detectable levels of particular norovirus strains were then incubated with the magnetic beads. After extensive washing to remove unbound virus, the magnetic beads were 10-fold serially diluted and subjected to heat release to extract the RNA genome. The virus-bead suspensions were diluted for two reasons: (i) to generate a semiquantitative result of viral attachment and (ii) to dilute any possible inhibitors of the RT-PCR amplification. Finally, RT-PCR was performed to assay for the presence of viral RNA to detect the attachment of the virus to the specific histo-blood group antigens. (B) Type 1 and 3 chain histo-blood group antigens used in this study are shown in bold type. Enzymatic activities required to produce the various carbohydrates are shown in italics. H type 2 antigen, which is not present in high concentrations on the superficial surfaces of the gut (47), was also used in this study as a tissue-specific control.
FIG. 2.
Histo-blood group antigen attachment specificities of noroviruses obtained from clinical specimens. RT-PCR-positive products indicate attachment of norovirus strains to the specific synthetic histo-blood group antigens shown. RNA samples extracted from the same clinical stool specimens subjected to the attachment assay were used as positive controls (lane +), and sterile water was used for negative controls (lane −). “No Carbohydrate” represents mock-coated magnetic beads as a negative control for nonspecific viral binding.
Also consistent with our previous studies using VLP reagents (27), different norovirus strains exhibited a variety of histo-blood group antigen attachment phenotypes (Fig. 2). A second GI strain, M23, representative of the Desert Shield/Stavanger-virus genotype (GI.3) did not attach to any of the histo-blood group antigens tested. This observation was confirmed by repeating the experiment with a 10-fold-higher concentration of virus (data not shown). Two GII strains, M15 and M116, bound specifically only to Leb. The M15 virus is representative of the Toronto (GII.3) virus genotype, while M116 represents a novel norovirus genotype (52).
Finally, SMV, which is a prototype GII strain (GII.2), was obtained from a human challenge stool specimen and tested for attachment to the various histo-blood group antigens. Previous studies from our laboratory demonstrated that SMV VLPs did not attach to any of the synthetic carbohydrates used in this study (27). However, we observed that SMV from the clinical stool specimen did in fact attach specifically to H type 3 (Fig. 2).
SMV attachment characterization.
We further investigated the SMV-H type 3 attachment properties to identify the cause of the discrepancy between the SMV VLP and clinical virus attachment observations. First, we tested if SMV VLPs will compete with the virus for attachment to H type 3. Using the magnetic bead method described above, the SMV stool specimen was added to H type 3-coated magnetic beads in either the absence or presence of excess SMV VLPs (10 μg, sucrose pure) or to beads pretreated with the same amount of SMV VLPs and then washed with PBS before addition of the SMV stool specimen. Interestingly, SMV VLPs outcompeted the virus to attach to H type 3 (Fig. 3). However, the SMV VLPs could be washed from the H type 3-coated beads before addition of the SMV stool specimen, restoring the ability of the virus to attach to H type 3 (Fig. 3).
FIG. 3.
Attachment of wild-type SMV to H type 3 in the presence and absence of VLPs. SMV from a clinical stool specimen was added to H type 3-coated magnetic beads and assayed for attachment by RT-PCR as described in the legend to Fig. 1. The effects of adding excess sucrose-purified SMV VLPs (10 μg) to the attachment reaction mixture (lanes +SMV VLPs) or preincubating and then washing the VLPs prior to adding the SMV clinical stool specimen (lanes + SMV VLPs, washed) were investigated.
We then used a series of microwell-based assays to further characterize SMV VLP attachment to H type 3. Using a biochemical microwell method, which previously established that SMV VLPs do not attach to H type 3 (27), we first characterized the attachment of three additional batches of sucrose-purified SMV VLPs. Consistent with the previous results, synthetic, biotinylated H type 3 did not attach to microwells coated with any of the three different batches of SMV VLPs (data not shown).
Using another method to detect VLP attachment to carbohydrate-coated microwells, similar to the method previously described by Hutson et. al. (33), SMV VLPs again did not attach to H type 3-coated microwells (Fig. 4). However, when the SMV stool specimen was added to sample buffer along with the VLPs, attachment to H type 3 was detected (Fig. 4). The binding signal appeared in a linear fashion in relation to VLP concentration, suggesting that the SMV VLPs and not the SMV virions in stool attached to H type 3.
FIG. 4.
Attachment of SMV VLPs to H type 3. The ability of SMV VLPs to attach to H type 3 in the presence or absence of the SMV clinical stool extract was investigated by using a microwell-based attachment assay. Wells were coated with H type 3 and blocked overnight, and the indicated protein concentrations of SMV VLPs (with or without 1% [vol/vol] stool extract in the sample buffer) were added. The wells were then washed, and VLP attachment was quantified by using human polyclonal anti-norovirus antiserum, goat anti-human IgG-alkaline phosphatase conjugate, and pNPP substrate. Net OD 405nm represents the OD405 values corrected for blank OD405 values (no VLPs added). Mean values from triplicate wells are shown, and error bars represent the standard deviation.
Stool contents promote SMV attachment to H type 3.
To determine if SMV VLP attachment is dependent on the presence of SMV virions or on another component of the extracellular environment in the clinical sample, we investigated the ability of other stool extracts to enhance SMV VLP attachment to H type 3. Forty-two archived clinical stool specimens from various norovirus outbreaks, representing at least 10 different norovirus genotypes, were screened for their ability to enhance SMV VLP attachment to H type 3. Remarkably, nearly all of the extracts promoted SMV VLP attachment to some extent (Fig. 5). The mean attachment (net OD405 ± standard deviation) of SMV VLPs to H type 3 in the presence of the stool samples was 1.57 ± 0.84, whereas attachment of three different batches of SMV VLPs to H type 3 in the absence of stool samples was 0.08 ± 0.01. The difference in the ability of SMV VLPs to attach to H type 3 in the presence and absence of stool extracts is significant (P < 0.001; Student's t test). No correlation was found between attachment enhancement levels and the presence, absence, genotype, or titer of noroviruses in the outbreak specimens (data not shown). In fact, even prechallenge, virus-free stool extracts from human volunteers promoted SMV attachment regardless of the individuals' secretor phenotype (Fig. 5). Therefore, stool contents alone are most likely promoting SMV attachment to H type 3, suggesting that a component of the extracellular environment in normal human feces might play a role in SMV attachment in vivo.
FIG. 5.
Effect of nonspecific stool extracts on SMV VLP attachment to H type 3. The effect of various archived stool extracts on SMV VLP attachment (10 μg/ml) to H type 3 was investigated by using the same microwell-based assay described in the legend to Fig. 4. Lanes Se+ Pre-challenge and Se− Pre-challenge contain virus-free stool extracts from secretor-positive and secretor-negative human volunteers, respectively, obtained prior to norovirus challenge.
We further investigated the attachment enhancement mechanism to characterize its specificity for SMV and H type 3. The ability of the SMV stool sample to enhance SMV VLP attachment was specific for H type 3, since attachment to H type 1, the H type 3 precursor, or mock-coated microwells was not detected even in the presence of the stool specimen (Fig. 6). In fact, neither wild-type SMV nor SMV VLPs attached to any of the carbohydrates used in this study other than H type 3, regardless of the presence or absence of stool extracts (Fig. 2 and data not shown).
FIG. 6.
SMV VLP attachment to other histo-blood group antigens in the presence of stool extract. Attachment of SMV VLPs in the presence of stool extract was investigated using the same microwell-based assay described in the legends to Fig. 4 and 5. A pool of no. 22, no. 27 (numbers are shown in Fig. 5), and SMV stool extracts was used at a final concentration of 1% in the sample buffer when added with the VLPs. Mean OD405 values from duplicate wells are shown, and error bars represent the standard deviation.
The attachment enhancement mechanism was not necessarily specific for SMV. The ability of NV VLPs to attach to H type 1, H type 3, or Leb was not enhanced by the presence of stool contents (Fig. 7A). In addition, no enhancement effect on NV VLP attachment with other histo-blood group antigens was evident (data not shown). However, we previously demonstrated that HV VLPs do not attach to any of the histo-blood group antigens used in this study (27). Although the effect was not as robust as for the SMV VLPs, addition of stool extracts modestly promoted HV VLP attachment specifically to Leb (Fig. 7B).
FIG. 7.
Effect of stool components on NV and HV VLP attachment to histo-blood group antigens. (A) The effect of stool on NV VLP attachment to H type 1, H type 3, and Leb was investigated using the same microwell-based assay described in the legends to Fig. 4 and 5. Note that different “1×” VLP concentrations of 0.2 μg/ml for H type 1 and 0.4 μg/ml for H type 3 and Leb were used to account for previously determined differences in attachment avidity of the different carbohydrates with NV VLPs (27). (B) Effect of stool extract on HV VLP attachment. Data shown are based on using 10 μg of HV VLP per ml. The dashed line represents an arbitrary background cutoff equivalent to twice the average background OD405. In all assays, a pool of no. 22, no. 27 (numbers from fig. 5), and SMV stool extracts was used at a final concentration of 1% in the sample buffer when added with the indicated VLPs.
We further characterized the attachment enhancement mechanism to begin to dissect the components of stool that might play a role. Enzyme-linked immunosorbent assays of the stool extracts were performed to determine if antibody might play a role in SMV attachment enhancement. Of the 42 different outbreak specimens tested, only a small fraction had detectable levels of norovirus-specific IgA, IgM, or IgG, with no correlation between the presence of detectable norovirus-specific antibody and the ability of the stool extracts to enhance SMV attachment (data not shown). In fact, denaturing the stool samples by boiling did not impair their ability to enhance SMV attachment to H type 3 (Fig. 8), suggesting that neither antibody, enzymes, nor other protein components in stool are responsible for SMV attachment enhancement.
FIG. 8.
Effect of denaturing stool extracts on SMV VLP attachment to H type 3. Stool extracts (no. 22, no. 27 [numbers from fig. 5], and SMV) were denatured by boiling for 10 min before being added to the sample buffer with SMV VLPs (10 μg of VLPs per ml plus 1% stool extract). Mean OD405 values for SMV VLP attachment in the presence of each of the three different extracts are shown, and error bars represent the standard deviation.
We then determined if pretreatment of the SMV VLPs or H type 3 with the stool extracts was sufficient to detect the attachment of SMV VLPs to H type 3. Before the addition of SMV VLPs, H type 3-coated microwells were pretreated with stool extracts for 2 h and washed to remove unbound stool components. Similarly, SMV VLP-coated microwells were pretreated with stool components and washed before addition of synthetic H type 3. In both cases, SMV VLP attachment to H type 3 was evident only when all three components—stool extract, SMV VLPs, and carbohydrate—were present at the same time (Fig. 9). Pretreatment of the SMV VLPs or H type 3 with stool extracts was not sufficient to promote attachment.
FIG. 9.
Effect of pretreating VLPs or carbohydrate with stool extracts followed by washing on SMV VLP attachment to H type 3. (A) SMV VLPs were serially diluted and added to H type 3-coated microwells in either the absence or presence of 1% stool extracts or to coated wells that were pretreated for 2 h with sample buffer containing 1% stool extracts followed by washing before the addition of the VLPs. VLP attachment was quantified as described in the legends to Fig. 4 and 5. (B) Biotinylated H type 3 carbohydrates were serially diluted and added to SMV VLP-coated microwells in either the presence or absence of 1% stool extracts or to coated wells that were pretreated for 2 h with sample buffer containing 1% stool extracts followed by washing before the addition of adding biotinylated H type 3. Attachment of biotinylated H type 3 to SMV VLPs was quantified by adding streptavidin-alkaline phosphatase conjugate and pNPP substrate. Net OD 405nm indicates OD405 values corrected for plate blank values (no H type 3 added). In both experiments, mean OD405 values for attachment in the presence or absence of three different stool extracts (no. 22, no. 27 [numbers from Fig. 5], or SMV) are shown, and error bars represent the standard deviation.
DISCUSSION
Several recent studies by multiple investigators have implicated the histo-blood group antigens as putative receptors for NV (27, 31, 33, 42). The importance of these molecules in vivo has been established using a human challenge model for NV infection in which secretor-negative individuals were resistant to live NV challenge (39). We have also previously observed three distinct patterns of binding, both to specific histo-blood group antigens and to histo-blood group antigens in saliva, for three different norovirus strains (27). These initial observations were supported by another study that revealed three potential additional binding patterns to saliva samples (31), although saliva-binding studies alone cannot distinguish which specific molecules are involved in attachment. Taken together, these observations suggest that the ABH histo-blood group antigens are important factors in norovirus pathogenesis and that various degrees of susceptibility may exist for different norovirus strains based on one's blood type, secretor phenotype, and/or Lewis phenotype.
To our knowledge, all studies prior to this utilized only VLP reagents to characterize norovirus attachment in vitro (27, 31, 33, 42, 50, 53). In this study, we designed a method to characterize the attachment of wild-type noroviruses obtained from clinical specimens with specific histo-blood group antigens to (i) determine if different attachment properties between VLPs and wild-type virus exist, (ii) reduce the need to produce additional VLP reagents to investigate the attachment properties of various norovirus strains, and (iii) develop an approach to concentrate and purify wild-type noroviruses from clinical or environmental samples. We found that different norovirus strains exhibited different histo-blood group antigen attachment patterns, consistent with previous studies using VLPs (27). However, in the case of SMV, wild-type virus bound to H type 3 whereas our previous study with SMV VLPs demonstrated no attachment to H type 3 (27).
Surprisingly, adding stool components to the attachment reaction mixtures promoted the specific attachment of SMV VLPs to H type 3, suggesting that the contradictory observations of SMV attachment are not due to phenotypic differences in wild-type virus and VLPs. In fact, excess amounts of SMV VLPs clearly outcompeted SMV virions for attachment to H type 3-coated magnetic beads, suggesting that both the virus and VLPs attach to H type 3 in a similar manner. This phenomenon was not specific for SMV-positive stool specimens since stool extracts from unrelated stool samples also efficiently promoted the attachment of SMV VLPs to H type 3 in microwell-based assays. Because noroviruses are typically transmitted by the fecal-oral route, the findings that both SMV and HV VLPs attached to histo-blood group antigens only in the presence of fecal extracts suggest that one or more stool components, as elements of the extracellular environment, might play a role in norovirus attachment in vivo. We therefore speculate that identification of this attachment enhancement factor could potentially lead to a target for novel, broadly reactive norovirus treatments and/or vaccines.
It is well established that the infectivity of many viral pathogens is enhanced by components of the extracellular environment. An extracellular proteolytic event that probably occurs in the intestine enhances the infectivity of several other enteric viruses, such as rotaviruses and reoviruses, probably at the level of attachment and entry (3, 6-8, 12, 13). Also, the infectivity of certain other viral pathogens, such as Dengue virus, is enhanced by the presence of virus-specific antibody (26, 44, 49).
It is unclear which particular component of stool might be promoting or enhancing norovirus attachment to histo-blood group antigens. Our results suggest that antibody, enzymes, and other protein molecules are not playing any role since denaturing the stool extracts did not impair their ability to enhance SMV VLP attachment to H type 3. In addition, extracts obtained from secretor-negative and secretor-positive individuals enhanced SMV attachment equally, suggesting that histo-blood group antigens secreted from the gut are not involved in promoting SMV attachment to H type 3. It is also puzzling that the stool extracts must be present with both the SMV VLPs and H type 3 to detect attachment, which suggests that the component is unlikely to be a ligand that first attaches to the virus and then links it to the carbohydrate, or vice versa. The acidity of the stool extracts is probably not playing a role, since the pH of the sample buffers used in the attachment assays does not change with the addition of stool extracts (data not shown). Divalent cations in stool extracts do not appear to play a role, since addition of MgCl2 and CaCl2 or EDTA to the sample buffers does not remarkably alter SMV VLP attachment properties (unpublished data). Other possible factors not ruled out in these studies include various salts, lipids, peptides, and proteins resistant to heat denaturing and other carbohydrate molecules that could be present as components of stool. Future experiments designed to fractionate the extracts by high-performance liquid chromatography or other methods will be conducted to attempt to precisely identify the factor(s) involved in promoting or enhancing norovirus attachment.
A similar observation of fecal or intestinal contents playing a role in viral infectivity has been made with an enteric calicivirus of pigs. Porcine enteric calicivirus Cowden (PEC) is the only human or animal enteric calicivirus known to replicate in cell culture (19). Interestingly, the virus is cultivable only when porcine intestinal contents are added to the medium (19, 46). Unlike SMV and HV, however, porcine intestinal contents probably do not affect the propagation of PEC in tissue culture at the level of viral attachment. Rather, a component of the intestinal contents is probably acting on cell signaling machinery to promote PEC replication (11). Nevertheless, the fact that fecal or intestinal contents affect the biology of multiple caliciviruses warrants further investigation of the identity and the role of environmental components in calicivirus attachment, infectivity, replication, and pathogenesis. Identifying and optimizing the use of such components could be essential in the development of a tissue culture or animal model system for cultivating human noroviruses, as in the case of PEC.
The magnetic bead-based technique for characterizing norovirus attachment described in this study not only provided a method to map histo-blood group antigen attachment properties but also was central in the observation that stool components enhance SMV attachment. Therefore, we propose that additional experiments in mapping the attachment properties of different norovirus strains should include methods to directly study viruses in stool, used either alone or in combination with previously described VLP-based methods. Addressing norovirus attachment questions using VLP reagents alone might result in the overlooking of molecules that are recognized for attachment only by wild-type virus in its more natural environment. This point is clearly illustrated in the fact that our previous studies with SMV VLPs failed to identify H type 3 as a possible attachment molecule (27). In addition, the methods described in this paper will be useful in identifying other norovirus strains that attach to histo-blood group antigens only in the presence of stool components or in identifying additional environmental components that influence norovirus attachment.
On a more practical level, these methods could also be valuable for concentrating, purifying, or diagnosing noroviruses in clinical or environmental samples to aid in norovirus molecular biology, epidemiology, and environmental science studies. In addition, with a more complete picture of norovirus-histo-blood group antigen interactions, the methods described in this paper might be useful for detecting and quantifying infectious virus. Unlike conventional RT-PCR diagnostics and previously described antibody-based norovirus capture methods (20), capture with histo-blood group antigen-coated magnetic beads selects for intact virions that are capable of attaching to these ligands, potentially providing a more accurate depiction of infectious viruses in the samples.
Future experiments will be aimed at mapping the histo-blood group antigen attachment patterns of noroviruses representing all of the major genogroups and genotypes. However, several limitations of the magnetic bead-based method exist, and will have to be addressed before all the different norovirus histo-blood group antigen binding patterns can be elucidated. One assay limitation is that high titers of the norovirus strains are required to detect attachment to the carbohydrate-coated beads. Although we can detect NV attachment to H type 1 over three 10-fold serial dilutions of the beads, addition of 1/10 of the clinical sample resulted in no detection of NV to H type 1, H type 3, or Leb, regardless of the bead dilution (data not shown). Therefore, only norovirus clinical samples that have a titer of at least 105 RT-PCR-detectable units per ml of stool suspension could be used in this assay in its present format, which screens out most norovirus-positive outbreak specimens. For this reason, although equal amounts of RT-PCR-detectable virus were added in the experiments, we cannot completely rule out the possibility of obtaining false-negative results for attachment. Our laboratory is currently investigating several potential solutions to this problem, including concentrating viruses using the magnetic-bead approach, reducing the total volume used for the assay, using new-generation streptavidin-coated beads, increasing the overall sensitivity of the RT-PCR amplification by using nested or real-time RT-PCR methods, and designing a flow cytometry, antibody-based method to detect viral attachment to carbohydrate-coated beads. Another potential problem is the presence of RT- PCR inhibitors in the reaction mixtures that remain after heat release of the RNA genome, especially in undiluted virus-bead suspensions. Although it would add several additional steps to the method, chemical extraction of the RNA genomes may increase the overall sensitivity of the assay.
Despite these limitations, several novel observations of norovirus attachment have been revealed in this study. A summary of the norovirus attachment data compiled with results from previous studies is shown in Fig. 10. No obvious histo-blood group antigen binding pattern based on genetic identity seems to exist, although none of the sequences analyzed between strains tested in this study were more than 75% identical. It is of interest that only the histo-blood group antigens that would be present in secretor-positive individuals were involved in attachment of the noroviruses analyzed in this study. We therefore speculate that secretor-negative individuals might be genetically resistant to a variety of norovirus strains, not just NV. However, most adult humans have serum antibody against noroviruses regardless of their secretor status (14, 16, 39, 45; C. Moe et al., unpublished data), suggesting that some norovirus strains might be able to infect secretor-negative individuals. In support of these observations, VLPs of one particular norovirus stain (VA207) bound to saliva from secretor-negative individuals (31), although the VLPs also bound modestly to saliva from secretor-positive individuals. Importantly, it remains unclear whether noroviruses utilize histo-blood group antigens as primary receptor molecules, coreceptors, or attachment-enhancing molecules or if all norovirus strains even attach to histo-blood group antigens.
It is also important to acknowledge that data from in vitro attachment experiments alone do not directly address whether one particular population would be more resistant or susceptible to norovirus infection and that these hypotheses are based only on the results obtained from human challenge studies of NV susceptibility (32, 39). Only human challenge experiments with other norovirus strains will provide results to conclusively resolve the role of histo-blood group antigens in human susceptibility to other noroviruses. However, results of attachment studies will allow the proposal of novel hypotheses of human susceptibility for other noroviruses, which can then be addressed using the human challenge model. For example, our data demonstrate that noroviruses from the M116 and M15/Toronto genotypes can utilize Leb as an attachment molecule. We therefore hypothesize that Lewis-negative and/or secretor-negative individuals might be more resistant to these norovirus strains than Lewis-positive and secretor-positive individuals, since either “negative” phenotype will result in the lack of Leb production on mucosal cell surfaces of the gut. Of course, the logistics of norovirus human challenge experiments do not conveniently allow for challenge with many different norovirus strains. Epidemiology studies, however, could address these questions to provide some information about the role of histo-blood group antigens in the infectivity of other norovirus strains. In a recent report by Hennessy et al. (30), blood group O individuals appeared to be more susceptible to infection during a norovirus outbreak that occurred in a closed military hospital setting, providing indirect evidence of the role of histo-blood group antigens in the infectivity of this particular strain. Therefore, with the combination of human challenge experiments, epidemiological observations, and in vitro attachment studies, human susceptibility to a variety of norovirus strains can be systematically investigated.
Identification of additional norovirus histo-blood group antigen binding patterns could also provide valuable insight into norovirus epidemiology and evolution. For example, the ability of noroviruses to bind to particular histo-blood group antigens could play a role in the establishment of norovirus strains selected and optimized to infect particular human populations, thus driving norovirus evolution. In fact, VLPs of Lordsdale virus, which is the genotype that has caused the majority of norovirus epidemics in recent years (9, 17), attach to at least five different specific histo-blood group antigens, listed here from highest to lowest binding avidity: H type 3, blood group B Ag, blood group A antigen, Leb, and H type 1 (unpublished data). We speculate this particular strain has evolved to utilize a greater variety of histo-blood group antigens for attachment to enhance its infectivity in a broad range of human populations.
In summary, this study has built on earlier observations of norovirus attachment and has provided the first examination of the attachment properties of wild-type noroviruses obtained from clinical stool specimens. In addition, we observed that stool extracts could promote norovirus attachment, suggesting that a component of the extracellular environment might play a role in enhancing norovirus attachment and infectivity in vivo.
Acknowledgments
We thank Martha Collier for producing packaged Venezuelan equine encephalitis VRPs for this study and Sjon Oudejans, Anna LoBue, Lisa Lindesmith, and Boyd Yount for helpful discussions and technical assistance.
This work was supported by grants from the U.S. Environmental Protection Agency (STAR grant R- 826139 and R-82936501) and the National Institutes of Health (AI056351, AI23946, and GM63228).
REFERENCES
- 1.Ando, T., J. S. Noel, and R. L. Fankhauser. 2000. Genetic classification of “Norwalk- like viruses”. J. Infect. Dis. 181(Suppl. 2):S336-S348. [DOI] [PubMed] [Google Scholar]
- 2.Arness, M. K., B. H. Feighner, M. L. Canham, D. N. Taylor, S. S. Monroe, T. J. Cieslak, E. L. Hoedebecke, C. S. Polyak, J. C. Cuthie, R. L. Fankhauser, C. D. Humphrey, T. L. Barker, C. D. Jenkins, and D. R. Skillman. 2000. Norwalk-like viral gastroenteritis outbreak in U.S. Army trainees. Emerg. Infect. Dis. 6:204-207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Babiuk, L. A., K. Mohammed, L. Spence, M. Fauvel, and R. Petro. 1977. Rotavirus isolation and cultivation in the presence of trypsin. J. Clin. Microbiol. 6:610-617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ball, J. M., M. E. Hardy, R. L. Atmar, M. E. Conner, and M. K. Estes. 1998. Oral immunization with recombinant Norwalk virus-like particles induces a systemic and mucosal immune response in mice. J. Virol. 72:1345-1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baric, R. S., B. Yount, L. Lindesmith, P. R. Harrington, S. R. Greene, F. C. Tseng, N. Davis, R. E. Johnston, D. G. Klapper, and C. L. Moe. 2002. Expression and self- assembly of norwalk virus capsid protein from venezuelan equine encephalitis virus replicons. J. Virol. 76:3023-3030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barnett, B. B., R. S. Spendlove, and M. L. Clark. 1979. Effect of enzymes on rotavirus infectivity. J. Clin. Microbiol. 10:111-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bass, D. M., D. Bodkin, R. Dambrauskas, J. S. Trier, B. N. Fields, and J. L. Wolf. 1990. Intraluminal proteolytic activation plays an important role in replication of type 1 reovirus in the intestines of neonatal mice. J. Virol. 64:1830-1833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bodkin, D. K., M. L. Nibert, and B. N. Fields. 1989. Proteolytic digestion of reovirus in the intestinal lumens of neonatal mice. J. Virol. 63:4676-4681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Centers for Disease Control and Prevention. 2003. Norovirus activity—United States, 2002. Morb. Mortal. Wkly. Rep. 52:41-45. [PubMed] [Google Scholar]
- 10.Centers for Disease Control and Prevention. 2002. Outbreaks of gastroenteritis associated with noroviruses on cruise ships—United States, 2002. Morb. Mortal. Wkly. Rep. 51:1112-1115. [PubMed] [Google Scholar]
- 11.Chang, K. O., Y. Kim, K. Y. Green, and L. J. Saif. 2002. Cell-culture propagation of porcine enteric calicivirus mediated by intestinal contents is dependent on the cyclic AMP signaling pathway. Virology 304:302-310. [DOI] [PubMed] [Google Scholar]
- 12.Clark, S. M., J. R. Roth, M. L. Clark, B. B. Barnett, and R. S. Spendlove. 1981. Trypsin enhancement of rotavirus infectivity: mechanism of enhancement. J. Virol. 39:816-822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Crawford, S. E., S. K. Mukherjee, M. K. Estes, J. A. Lawton, A. L. Shaw, R. F. Ramig, and B. V. Prasad. 2001. Trypsin cleavage stabilizes the rotavirus VP4 spike. J. Virol. 75:6052-6061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cubitt, W. D., K. Y. Green, and P. Payment. 1998. Prevalence of antibodies to the Hawaii strain of human calicivirus as measured by a recombinant protein based immunoassay. J. Med. Virol. 54:135-139. [PubMed] [Google Scholar]
- 15.De Leon, R., S. M. Matsui, R. S. Baric, J. E. Herrmann, N. R. Blacklow, H. B. Greenberg, and M. D. Sobsey. 1992. Detection of Norwalk virus in stool specimens by reverse transcriptase-polymerase chain reaction and nonradioactive oligoprobes. J. Clin. Microbiol. 30:3151-3157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dimitrov, D. H., S. A. Dashti, J. M. Ball, E. Bishbishi, K. Alsaeid, X. Jiang, and M. K. Estes. 1997. Prevalence of antibodies to human caliciviruses (HuCVs) in Kuwait established by ELISA using baculovirus-expressed capsid antigens representing two genogroups of HuCVs. J. Med. Virol. 51:115-118. [PubMed] [Google Scholar]
- 17.Fankhauser, R. L., S. S. Monroe, J. S. Noel, C. D. Humphrey, J. S. Bresee, U. D. Parashar, T. Ando, and R. I. Glass. 2002. Epidemiologic and molecular trends of “Norwalk-like viruses” associated with outbreaks of gastroenteritis in the United States. J. Infect. Dis. 186:1-7. [DOI] [PubMed] [Google Scholar]
- 18.Fankhauser, R. L., J. S. Noel, S. S. Monroe, T. Ando, and R. I. Glass. 1998. Molecular epidemiology of “Norwalk-like viruses” in outbreaks of gastroenteritis in the United States. J. Infect. Dis. 178:1571-1578. [DOI] [PubMed] [Google Scholar]
- 19.Flynn, W. T., and L. J. Saif. 1988. Serial propagation of porcine enteric calicivirus-like virus in primary porcine kidney cell cultures. J. Clin. Microbiol. 26:206-212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gilpatrick, S. G., K. J. Schwab, M. K. Estes, and R. L. Atmar. 2000. Development of an immunomagnetic capture reverse transcription-PCR assay for the detection of Norwalk virus. J. Virol. Methods 90:69-78. [DOI] [PubMed] [Google Scholar]
- 21.Glass, P. J., L. J. White, J. M. Ball, I. Leparc-Goffart, M. E. Hardy, and M. K. Estes. 2000. Norwalk virus open reading frame 3 encodes a minor structural protein. J. Virol. 74:6581-6591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Glass, R. I., J. Noel, T. Ando, R. Fankhauser, G. Belliot, A. Mounts, U. D. Parashar, J. S. Bresee, and S. S. Monroe. 2000. The epidemiology of enteric caliciviruses from humans: a reassessment using new diagnostics. J. Infect. Dis. 181(Suppl. 2):S254-S261. [DOI] [PubMed] [Google Scholar]
- 23.Green, J., J. Vinje, C. I. Gallimore, M. Koopmans, A. Hale, D. W. Brown, J. C. Clegg, and J. Chamberlain. 2000. Capsid protein diversity among Norwalk-like viruses. Virus Genes. 20:227-236. [DOI] [PubMed] [Google Scholar]
- 24.Green, K. Y., A. Z. Kapikian, and R. M. Chanock. 2001. Human caliciviruses, p. 841-874. In D. M. Knipe, P. M. Howley, D. E. Griffin, et al. (ed.), Fields virology, 4th ed. Lippincott-Raven, Philadelphia, Pa.
- 25.Guerrero, R. A., J. M. Ball, S. S. Krater, S. E. Pacheco, J. D. Clements, and M. K. Estes. 2001. Recombinant norwalk virus-like particles administered intranasally to mice induce systemic and mucosal (fecal and vaginal) immune responses. J. Virol. 75:9713-9722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Halstead, S. B. 1988. Pathogenesis of dengue: challenges to molecular biology. Science 239:476-481. [DOI] [PubMed] [Google Scholar]
- 27.Harrington, P. R., L. Lindesmith, B. Yount, C. L. Moe, and R. S. Baric. 2002. Binding of Norwalk virus-like particles to ABH histo-blood group antigens is blocked by antisera from infected human volunteers or experimentally vaccinated mice. J. Virol. 76:12335-12343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Harrington, P. R., L. Lindesmith, B. Yount, C. L. Moe, J. LePendu, and R. S. Baric. 2003. Norovirus attachment, susceptibility, and vaccine design. Recent Res. Dev. Virol. 5:19-44. [Google Scholar]
- 29.Harrington, P. R., B. Yount, R. E. Johnston, N. Davis, C. Moe, and R. S. Baric. 2002. Systemic, mucosal, and heterotypic immune induction in mice inoculated with Venezuelan equine encephalitis replicons expressing Norwalk virus-like particles. J. Virol. 76:730-742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hennessy, E. P., A. D. Green, M. P. Connor, R. Darby, and P. MacDonald. 2003. Norwalk virus infection and disease is associated with ABO histo-blood group type. J. Infect. Dis. 188:176-177. [DOI] [PubMed] [Google Scholar]
- 31.Huang, P., T. Farkas, S. Marionneau, W. Zhong, N. Ruvoen-Clouet, A. L. Morrow, M. Altaye, L. K. Pickering, D. S. Newburg, J. LePendu, and X. Jiang. 2003. Noroviruses bind to human ABO, Lewis, and secretor histo-blood group antigens: identification of 4 distinct strain-specific patterns. J. Infect. Dis. 188:19-31. [DOI] [PubMed] [Google Scholar]
- 32.Hutson, A. M., R. L. Atmar, D. Y. Graham, and M. K. Estes. 2002. Norwalk virus infection and disease is associated with ABO histo-blood group type. J. Infect. Dis. 185:1335-1337. [DOI] [PubMed] [Google Scholar]
- 33.Hutson, A. M., R. L. Atmar, D. M. Marcus, and M. K. Estes. 2003. Norwalk virus- like particle hemagglutination by binding to h histo-blood group antigens. J. Virol. 77:405-415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiang, X., M. Wang, D. Y. Graham, and M. K. Estes. 1992. Expression, self-assembly, and antigenicity of the Norwalk virus capsid protein. J. Virol. 66:6527-6532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jiang, X., M. Wang, K. Wang, and M. K. Estes. 1993. Sequence and genomic organization of Norwalk virus. Virology 195:51-61. [DOI] [PubMed] [Google Scholar]
- 36.Jiang, X., N. Wilton, W. M. Zhong, T. Farkas, P. W. Huang, E. Barrett, M. Guerrero, G. Ruiz-Palacios, K. Y. Green, J. Green, A. D. Hale, M. K. Estes, L. K. Pickering, and D. O. Matson. 2000. Diagnosis of human caliciviruses by use of enzyme immunoassays. J. Infect. Dis. 181(Suppl. 2):S349-S359. [DOI] [PubMed] [Google Scholar]
- 37.Karst, S. M., C. E. Wobus, M. Lay, J. Davidson, and H. W. t. Virgin. 2003. STAT1- dependent innate immunity to a Norwalk-like virus. Science 299:1575-1578. [DOI] [PubMed] [Google Scholar]
- 38.Koopmans, M., C. H. von Bonsdorff, J. Vinje, D. de Medici, and S. Monroe. 2002. Foodborne viruses. FEMS Microbiol Rev. 26:187-205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lindesmith, L., C. Moe, S. Marionneau, N. Ruvoen, X. Jiang, L. Lindblad, P. Stewart, J. LePendu, and R. Baric. 2003. Human susceptibility and resistance to Norwalk virus infection. Nat. Med. 9:548-553. [DOI] [PubMed] [Google Scholar]
- 40.Lopman, B. A., G. K. Adak, M. H. Reacher, and D. W. Brown. 2003. Two epidemiologic patterns of norovirus outbreaks: surveillance in England and Wales, 1992-2000. Emerg. Infect. Dis. 9:71-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Marionneau, S., A. Cailleau-Thomas, J. Rocher, B. Le Moullac-Vaidye, N. Ruvoen, M. Clement, and J. Le Pendu. 2001. ABH and Lewis histo-blood group antigens, a model for the meaning of oligosaccharide diversity in the face of a changing world. Biochimie 83:565-573. [DOI] [PubMed] [Google Scholar]
- 42.Marionneau, S., N. Ruvoen, B. Le Moullac-Vaidye, M. Clement, A. Cailleau- Thomas, G. Ruiz-Palacois, P. Huang, X. Jiang, and J. Le Pendu. 2002. Norwalk virus binds to histo-blood group antigens present on gastroduodenal epithelial cells of secretor individuals. Gastroenterology 122:1967-1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mead, P. S., L. Slutsker, V. Dietz, L. F. McCaig, J. S. Bresee, C. Shapiro, P. M. Griffin, and R. V. Tauxe. 1999. Food-related illness and death in the United States. Emerg. Infect. Dis. 5:607-625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Morens, D. M. 1994. Antibody-dependent enhancement of infection and the pathogenesis of viral disease. Clin. Infect. Dis. 19:500-512. [DOI] [PubMed] [Google Scholar]
- 45.Parker, S. P., W. D. Cubitt, X. J. Jiang, and M. K. Estes. 1994. Seroprevalence studies using a recombinant Norwalk virus protein enzyme immunoassay. J. Med. Virol. 42:146-150. [DOI] [PubMed] [Google Scholar]
- 46.Parwani, A. V., W. T. Flynn, K. L. Gadfield, and L. J. Saif. 1991. Serial propagation of porcine enteric calicivirus in a continuous cell line. Effect of medium supplementation with intestinal contents or enzymes. Arch. Virol. 120:115-122. [DOI] [PubMed] [Google Scholar]
- 47.Ravn, V., and E. Dabelsteen. 2000. Tissue distribution of histo-blood group antigens. Apmis 108:1-28. [DOI] [PubMed] [Google Scholar]
- 48.Smiley, J. R., K. O. Chang, J. Hayes, J. Vinje, and L. J. Saif. 2002. Characterization of an enteropathogenic bovine calicivirus representing a potentially new calicivirus genus. J. Virol. 76:10089-10098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sullivan, N. J. 2001. Antibody-mediated enhancement of viral disease. Curr. Top. Microbiol. Immunol. 260:145-169. [DOI] [PubMed] [Google Scholar]
- 50.Tamura, M., K. Natori, M. Kobayashi, T. Miyamura, and N. Takeda. 2000. Interaction of recombinant norwalk virus particles with the 105-kilodalton cellular binding protein, a candidate receptor molecule for virus attachment. J. Virol. 74:11589-11597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vinje, J., J. Green, D. C. Lewis, C. I. Gallimore, D. W. Brown, and M. P. Koopmans. 2000. Genetic polymorphism across regions of the three open reading frames of “Norwalk-like viruses”. Arch. Virol. 145:223-241. [DOI] [PubMed] [Google Scholar]
- 52.Vinje, J., R. A. Hamidjaja, and M. D. Sobsey. 2004. Development and application of a capsid VP1 (region D) based reverse transcription PCR assay for genotyping of genogroup I and II Noroviruses. J. Virol. Methods 109-117. 116: [DOI] [PubMed]
- 53.White, L. J., J. M. Ball, M. E. Hardy, T. N. Tanaka, N. Kitamoto, and M. K. Estes. 1996. Attachment and entry of recombinant Norwalk virus capsids to cultured human and animal cell lines. J. Virol. 70:6589-6597. [DOI] [PMC free article] [PubMed] [Google Scholar]