Abstract
Toll-like receptor 3 (TLR3) signaling is activated by double-stranded RNA (dsRNA), a virus-associated molecular pattern. Injection of dsRNA into mice induced a rapid, dramatic and reversible remodeling of the small intestinal mucosa with significant villus shortening. Villus shortening was preceded by increased caspase 3 and 8 activation and apoptosis of intestinal epithelial cells (IECs) located in the mid to upper villus with ensuing luminal fluid accumulation and diarrhea due to an increased secretory state. Mice lacking TLR3 or the adaptor molelcule TRIF mice were completely protected from dsRNA-induced IEC apoptosis, villus shortening and diarrhea. dsRNA induced apoptosis was independent of TNF signaling. Notably, NF-κB signaling through IκB kinase beta protected crypt IECs but did not protect villus IECs from dsRNA-induced or TNF-induced apoptosis. dsRNA did not induce early caspase 3 activation with subsequent villus shortening in mice lacking caspase 8 in IECs, but instead caused villus destruction with a loss of small intestinal surface epithelium and death. Consistent with direct activation of the TLR3-TRIF-caspase 8 signaling pathway by dsRNA in IECs, dsRNA-induced signaling of apoptosis was independent of non-TLR3 dsRNA signaling pathways, IL-15, TNF, IL-1, IL-6, IRF3, type I IFN receptor, adaptive immunity, as well as dendritic cells, NK cells, and other hematopoietic cells. We conclude that dsRNA activation of the TLR3-TRIF-caspase 8 signaling pathway in IECs has a significant impact on the structure and function of the small intestinal mucosa and suggest signaling through this pathway has a host protective role during infection with viral pathogens.
INTRODUCTION
The mucosa of the small intestine contains finger-like projections termed villi that are lined by a single-cell layer of intestinal epithelial cells (IECs) that separate the intestinal lumen from the underlying immune cell-rich lamina propria. IECs are armed with numerous pattern-recognition receptors (PRRs) that detect and activate host responses to protect against viral and bacterial pathogens and their products (1). Double-stranded RNA (dsRNA) is a virus-associated molecular pattern that is recognized by the endosomal PRR TLR3, and intracellular cytoplasmic RIG-like sensors (2, 3). Interestingly, dsRNA signaling through TLR3 has been reported to induce small intestinal mucosal damage in mice (4).
TLR3 signals through the adaptor molecule TRIF, whereas the RIG-like RNA sensors, RIG-I and Mda5, signal through the adaptor molecule MAVS (IPS-1) (2). These pathways activate IRF3 and NF-κB transcription factors and result in the production of type I interferons (IFNs) and proinflammatory cytokines. Furthermore, activation of host signaling pathways can induce apoptosis, likely as a means to kill infected host cells to shut down virus production (5). While NF-κB can have both pro- and anti-apoptotic effects within cells, IRF3 has been shown to interact with BAX to directly induce apoptosis after viral infection (6). Additionally, in vitro studies have shown that TRIF, unlike the TLR adaptor protein MyD88, interacts with caspase 8 through the RIP1 signaling molecule to directly induce apoptosis (7–10).
PRR signaling pathways can also induce TNF that can act upon other cells to either induce survival through NF-κB signaling or death through apoptosis or necroptosis (11). Interestingly, TNF injection induces IEC apoptosis in vivo in mice (12). This effect is dependent upon TNF receptor 1 (TNFR-1) and more pronounced in the proximal than distal small intestine, possibly due to the expression pattern of the TNFR-1 (13). Signaling through TNFR-1 leads to the recruitment of a signaling complex that activates caspase 8, which then mediates apoptosis by activating effector caspases including caspase 3 (11).
We investigated the mucosal signaling pathways activated by dsRNA that cause small intestinal damage in vivo. We report that dsRNA rapidly induces intestinal villus epithelial cell apoptosis and cell loss in the small intestine that results in marked villus shortening and significant diarrhea, with subsequent recovery. dsRNA-induced IEC apoptosis in vivo was strictly dependent on TLR3 and TRIF signaling through caspase 8 in IECs, and independent of other dsRNA signaling pathways, as well as TNF, IL-15, dendritic cells (DCs), natural killer (NK) cells, and other hematopoietic cells. Interestingly, this pathway proved to be host protective since, in the absence of downstream activation of caspase 8, dsRNA caused marked destruction of the small intestinal mucosa and death.
MATERIALS AND METHODS
Reagents
Low molecular weight pIC was from Invivogen (San Diego, CA) and used in all studies unless indicated otherwise. pIC sodium salt (S-pIC) was from Sigma-Aldrich (St. Louis, MO). R848, ODN 1826 and LPS from E. coli O111:B4 were from Invivogen. pIC was heated to 50°C for 10 minutes and allowed to cool to room temperature to anneal. Cleaved caspase 3 (Asp 175) and cleaved caspase 8 (Asp 387) (D5B2) antibody was from Cell Signaling (Danvers, MA). Recombinant mouse TNF was purchased from Peprotech (Rocky Hill, NJ).
Mouse strains and treatment
IEC-specific caspase-8 knockout mice (casp8ΔIEC) were generated in our laboratory by breeding Casp8fl mice (14) with villin-Cre mice (15) as described by Günther et al. (16). Wild type (WT) C57BL/6J, TNF−/−,TRIFlps2/lps2 (TRIF−/−) (17), BAX−/−(18), RAG1−/− (19), IL-6−/− (20) and IL-1R−/− (21) mice from The Jackson Laboratory, IL-15−/− mice (22) from Taconic, and IFNAR−/− (23), PKR−/− (24), TLR3−/− (25), CD11c-DTR (diphtheria toxin receptor) (26), IκK-βΔIEC (27), IκK-αΔIEC (28), TLR4−/− (29), IRF3−/− mice (30), MAVS−/− (31) and casp8ΔIEC mice were maintained in specific pathogen free conditions at UCSD. Mice were given a single intraperitoneal (i.p.) injection of 30 mg/kg pIC, 10 mg/kg R848, 5 mg/kg ODN 1826 or 20 mg/kg LPS in sterile phosphate buffered saline or saline solution, using sterile PBS or saline injection alone as a control. In one series of experiments pIC was injected in the retro-orbital sinus of anesthetized mice. Recombinant TNF was injected i.p. Mice were sacrificed at 3–6 h after injection unless otherwise indicated. All animal studies were approved by the UCSD Institutional Animal Care and Use Committee.
Histology and Immunohistochemistry
Proximal small intestine starting 2 cm distal to the pylorus and continuing for the first third of the small intestine was processed as Swiss rolls (32). Distal small intestine Swiss rolls were taken from the distal third of the small intestine. Tissues were fixed in 10% formalin and embedded in paraffin, and 5-µm sections were stained with H&E. For cleaved caspase 3 staining, samples were deparaffinized, subjected to antigen retrieval and incubated with antibody overnight, after which HRP conjugated secondary antibody was added for 2 h followed by incubation with 3,3'-diaminobenzidine (DAB) (Vector Laboratories) and counter-staining with hematoxylin. For NK1.1 and CD11c staining, small intestine Swiss rolls were frozen in OCT (Tissue-Tek). Frozen sections were fixed in neutral buffered formalin for 10 min, washed and blocked with 3% BSA in PBS. For NK1.1 staining, sections were incubated overnight with FITC-conjugated NK1.1 (PK136) (Abcam) or IgG negative control mAb then washed and visualized. For dendritic cell staining, sections were incubated overnight with CD11c antibody (BD Pharmingen) or Armenian hamster negative control (AbD Serotec) then washed and stained with Cy3 goat anti-Armenian hamster (Jackson Immunoresearch).
Microscopy analysis
Villus lengths were measured in each Swiss roll and the ten longest villi were averaged to give a final mean villus length. Cleaved caspase 3 stained cells that were completely black were counted as positive cells and ten representative villi were counted and averaged to provide a value for each mouse. Samples were examined in our laboratory under an Olympus BX41 microscope. Digital images were captured using the PictureFrame Application 2.3 (Optronics).
Ussing chamber studies
Segments of proximal small intestine were cut along the mesenteric border and mounted in Ussing chambers (Physiological instruments, San Diego, CA), exposing 0.09 cm2 of tissue area to 4 mL of circulating oxygenated Ringer’s buffer maintained at 37°C. The buffer consisted (in mM) of: 140 Na+, 5.2 K+, 1.2 Ca2+, 0.8 Mg2+, 120 Cl−, 25 HCO3−, 2.4 H2PO4−, 0.4 HPO42−. Additionally, glucose (10 mM) was added to the serosal buffer as a source of energy, osmotically balanced by mannitol (10 mM) in the mucosal buffer. Agar–salt bridges were used to monitor the potential difference across the tissue and to inject the required short-circuit current (Isc) to maintain the potential difference at zero. This was registered by an automated voltage clamp and continuously recorded by computer. Baseline Isc values were obtained at equilibrium, 15 min after the tissues were mounted and expressed as µA/cm2. Conductance (G) was also determined at baseline as an indicator of ion flux and expressed as mS/cm2. FITC labeled dextran (4 kDa, Sigma) was used as a probe to assess macromolecular permeability, and was added (2.2 mg/mL final concentration) to the luminal buffer once equilibrium was reached. Serosal samples (240 uL) were taken at 30 min intervals for 2 h and replaced with fresh buffer to maintain constant volume. Fluorescence was measured by end point assay (Victor4X, Perkin Elmer) and the flux of FITC-dextran from the mucosa to the serosa was calculated as the average value of two consecutive stable flux periods (60–90 and 90–120 min) and expressed as pmol/cm2/h. (33).
Cytokine detection
Serum samples were collected 1h after treatment and stored at −80°C. Cytokine measurements used the mouse ultra-sensitive TNF kit or proinflammatory 7-plex kit from Meso Scale Discovery (Gaithersburg, MD) and cytokine levels were determined using a Meso Scale Discover Sector Imager 6000.
RNA extraction and quantitative real-time PCR
Total cellular RNA was extracted using Trizol (Life Technologies, Carlsbad, CA), according to the manufacturer’s instructions. 1 µg RNA total RNA was used for iScript (BioRad Laboratories, Hercules, CA) reverse transcription and the cDNA was used for quantitative Real Time PCR (qPCR) using SYBR green Master mix (Applied Biosystems, Foster City, CA) and GAPDH forward primer ATCAACGACCCCTTCATTGACC and GAPDH reverse primer CCAGTAGACTCCACGAGATACTCAGC or IFN-β forward primer CTGAGCAGCTGAATGGAAAG and IFN-β reverse primer CTTGAAGTCCGCCCTGTAGGT. Denaturation was 5 min at 95°C followed by 40 cycles of amplification at 95°C for 30 s and 60°C for 30 s using an ABI StepOnePlus (Applied Biosystems). IFN-β induction was calculated using the ΔΔCt method and values were normalized to PBS treated mice.
Natural killer and dendritic cell depletion
To deplete NK cells, mice were injected with 200 µg purified anti-NK1.1 (PK136) mAb (eBioscience, San Diego, CA) or control IgG2 24 h before injection of pIC. NK1.1+ cell depletion was confirmed by immunohistochemistry as described above. For DC depletion, DTR (26) mice were treated with 4 ng/g diphtheria toxin i.p. (Sigma Aldrich) 24 h prior to pIC injection. DC depletion was confirmed by immunohistochemistry as described above.
Bone marrow chimeras
To generate bone marrow chimeric mice, WT or TLR3 recipients were exposed to a single lethal dose of 9 Gy total body irradiation from a 137Cesium source. Six hours later these mice were injected i.v. with 2–5×106 bone marrow cells from TLR3−/− or WT mice. Mice were rested for 8 weeks before use.
Statistical Analysis
Data are expressed as mean ± standard deviation. Two groups were compared using unpaired Student t test. Analysis of variance with Bonferonni post-test was used for multiple group comparisons. Significant differences were reported where p <0.05.
RESULTS
dsRNA induces IEC apoptosis in the small intestine
We used the synthetic dsRNA viral nucleic acid mimic polyinosinic:polycytidylic acid (pIC) as a probe of the host’s antiviral strategies in the small intestine. Administration of pIC i.p. resulted in villus shortening in the proximal small intestine, which was maximal between 3 and 6 h after injection and recovered thereafter (Fig. 1 A–C). The dramatic reduction in villus length correlated with decreased numbers of IECs per villus (Fig. S1 A–C). The relatively rapid loss of IECs and ensuing recovery suggested a dsRNA-activated pathway that causes IEC loss, perhaps as a host response to rid the epithelium of virus-infected cells, while concurrently maintaining a relatively intact IEC layer.
Figure 1. pIC induces IEC apoptosis and small intestinal villus shortening.
H&E stained sections of proximal WT mouse small intestine 3 h after (A) PBS or (B) pIC 30 µg/g i.p. Scale bar 200 µm. (C) Mean villus length at indicated times after pIC. (D) Cleaved caspase 3 stained sections of proximal small intestinal mucosa 2 h after (D) PBS or (E, F) pIC. (D, E) Scale bar 100 µm and (F) 25 µm. (G) Cleaved caspase 3 labeled cells per villus at specified times after pIC. Representative images of the peritoneal cavity of (H) PBS or (I) pIC 30 µg/g i.p. injected mice at 6 h. (J) Fluid weight per length of the entire small intestine at the indicated times after pIC. Values are mean ± SD, n=3–6 mice per time. ** p < 0.01 vs PBS. S = solid contents, no fluid. Ussing chamber measurements of (K) short circuit current (Isc), (L) conductance (G) and (M) macromolecular permeability to 4 kDa FITC-dextran in proximal small intestine pieces isolated from mice treated with PBS or 30 µg/g i.p. pIC for 3 h. Graphs show mean ± SEM, n = 5 samples per treatment group.
To determine if apoptosis explained the decreased numbers of IECs, we evaluated cleaved caspase 3 staining of small intestinal tissue sections (34) (Fig. 1 D–F). IEC apoptosis preceded villus shortening with a peak at 2 h after pIC i.p. (Fig. 1 G). Apoptotic IECs predominately localized in the mid to upper villus region, with little apoptosis in the lower villus region and no increase in apoptosis in either the crypt region (i.e., the site of epithelial stem cells) or the subepithelial lamina propria (Fig. 1 E). Cleavage of caspase 8, an initiator caspase, was also observed at 2 h post treatment (Fig. S1 D, E). Apoptosis of villus IECs and villus shortening were less pronounced but still significant in the distal small intestine (Fig. S1 F–I), likely reflecting shorter villi with decreased numbers of IECs in the distal small intestine.
pIC-treated mice had significant fluid accumulation in the small intestine and diarrhea that peaked at 4–6 h and returned to normal thereafter (Fig. 1 H–J). To address the cause of this increase in intestinal fluid accumulation, Ussing chamber studies were done to measure the small intestinal secretory state and mucosal permeability. Baseline Isc, indicative of active ion secretion, was elevated in mice treated with pIC compared to PBS controls (Fig. 1K). In contrast, conductance indicative of ion permeability (Fig. 1 L) and FITC-flux indicative of macromolecular permeability (Fig. 1 M) were not significantly altered by pIC treatment. Together, these data suggest that increased luminal fluid accumulation in the proximal small bowel resulted from an increased secretory state in these mice in the context of intact tight junctions and mechanisms of endocytosis.
We next investigated if the mode of pIC delivery determined IEC apoptosis, as injection by the i.p. route suggested the possible involvement of intraperitoneal macrophages or other cell subsets specific to the peritoneal cavity in inducing villus shortening and IEC apoptosis. However, there was no dependence on i.p. injection, as either i.p. or retro-orbital sinus injection of pIC induced significant villus shortening in the proximal small intestine and IEC apoptosis (Fig. S1 J–M, N and O). Moreover, Cy3-labeled pIC was detected in the lamina propria of proximal small intestinal villi after retro-orbital injection (Fig. S1 P–R). Administration of pIC by oral gavage did not induce villus shortening or IEC apoptosis (Fig. S1 M–O).
TLR3-TRIF signaling is required for IEC apoptosis
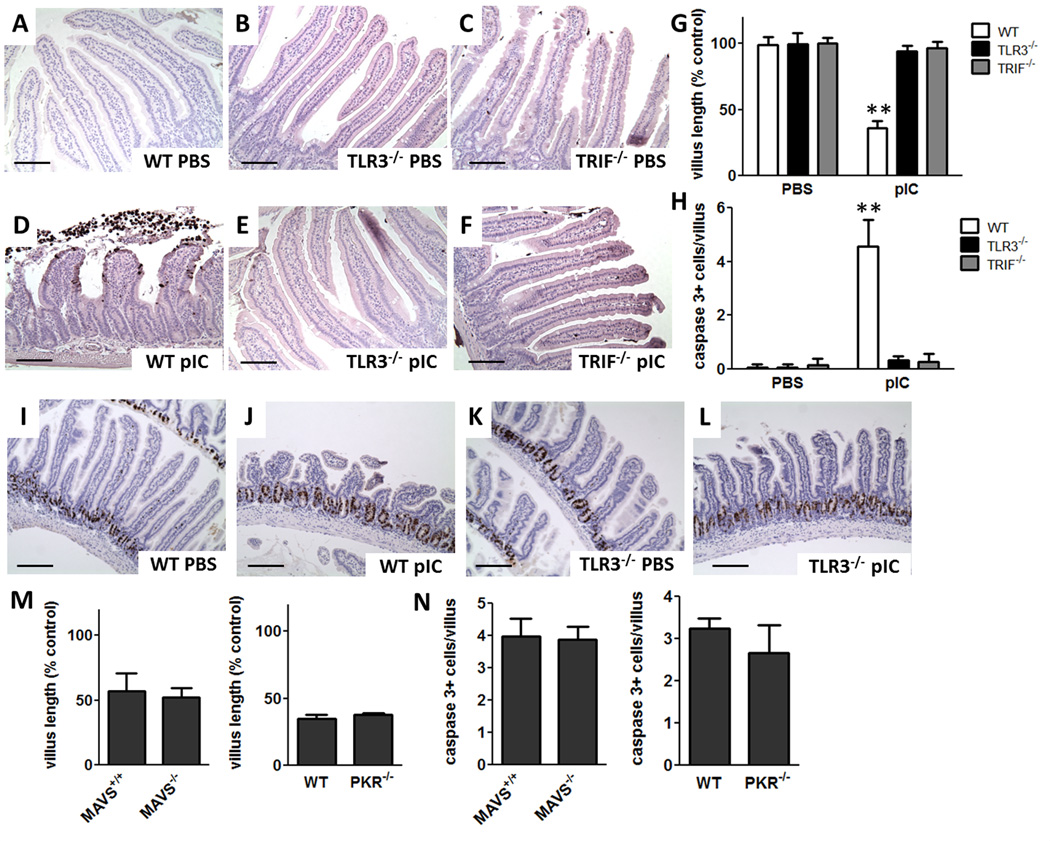
We found that TLR3 was required for dsRNA-induced intestinal damage as reported before by others (4). Indeed, TLR3−/− mice were completely resistant to dsRNA-induced IEC apoptosis and villus shortening (Fig. 2 A, B, D and E). Further, dsRNA did not induce IEC apoptosis or villus shortening in mice lacking TRIF, the downstream adaptor molecule for TLR3 (Fig. 2 C, F). Villus length and the number of IECs with cleaved caspase 3 staining per villus were quantified and confirmed the essential involvement of TLR3-TRIF signaling in pIC-activated villus shortening and apoptosis in the proximal small intestine (Fig 2 G and H). Neither TLR3−/− nor TRIF−/− mice developed small intestinal fluid accumulation or diarrhea. There was no difference in IEC proliferation between WT and TLR3−/− mice after pIC, indicating that pIC did not damage the proliferative capacity of crypt cells (Fig. 2 I–L).
Figure 2. TLR3-TRIF signaling is required for pIC-induced small intestinal villus shortening and IEC apoptosis.
Cleaved caspase 3 stained sections of proximal small intestine from (A–C) PBS and (D–F) 30 µg/g pIC i.p. treated WT, TLR3−/− and TRIF−/− mice at 3 h. Scale bar 100 µm. (G) Villus length and (H) cleaved caspase 3 labeled cells per villus after PBS or 30 µg/g pIC i.p. at 3 h. Data are mean ± SD, n=3–4 mice/group. ** p < 0.01 vs PBS treated mice and pIC treated TLR3−/− and TRIF−/− mice. BrdU stained sections from the proximal small intestine 6 h after (I) PBS i.p. or (J) 30 µg/g pIC i.p. in WT mice, (K) PBS i.p. or (L) 30 µg/g pIC i.p. in TLR3−/− mice. Scale bar 200 µm. (M) Villus length of MAVS+/+, MAVS−/−, WT and PKR−/− mice at 3 h after 30 µg/g pIC i.p.. (N) Cleaved caspase 3 labeled cells per villus for pIC treated MAVS+/+, MAVS−/−, WT and PKR−/− mice at 3 h after 30 µg/g pIC i.p.. Mean ± SD, n=5–8 mice/group.
We assessed the requirement for other RNA sensors to determine if they had a contributory role in pIC-activated IEC apoptosis in the small intestine. MAVS (IPS-1), the signaling adaptor for RIG-like receptors, was not required for villus shortening or caspase 3 cleavage (Fig. 2 M, N). PKR is an IFN-inducible protein kinase that can act as a general translation inhibitor and induce apoptosis in response to activation by dsRNA (35). Mice deficient in PKR had similar levels of villus shortening and numbers of caspase 3 cleaved cells as WT mice (Fig. 2 M, N) indicating that PKR-dependent translation inhibition or signaling was not required for pIC activated IEC apoptosis and villus shortening
Apoptosis of small intestinal IECs induced by TLR ligands
High levels of TNF result in IEC apoptosis (12) and, consistent with this, i.p. injection of 5 µg, but not 1 µg, of TNF induced IEC apoptosis in WT mice (Figs. 3 E–G). Since several TLR ligands that signal through MyD88 induce the release of TNF (2), we evaluated IEC apoptosis induced by other TLR ligands and found that pIC induced significantly higher levels of IEC apoptosis than the TLR7 ligand R848, TLR9 ligand ODN, and TLR4 ligand LPS (Figs. 3 A–D, G). Moreover, pIC induced TNF levels orders of magnitude lower than those TLR ligands, with LPS inducing the highest TNF levels (Fig. 3 H). While others observed higher serum TNF levels after pIC (4) we established that this was likely due to endotoxin contamination of pIC in their studies. However, we found such levels of endotoxin contamination did not significantly affect IEC apoptosis or villus shortening (Fig. S2).
Figure 3. pIC-induced IEC apoptosis is TNF independent.
Cleaved caspase 3 stained proximal small intestine sections from WT mice injected i.p. with (A) TLR3 ligand pIC, (B) TLR7 ligand R848, (C) TLR9 ligand ODN 1826, (D) TLR4 ligand LPS, (E) 5 µg TNF or (F) 1 µg TNF after 2 h. Scale bar 100 µm. (G) Cleaved caspase 3 labeled cells per villus in WT mouse proximal small intestine sections 3 h following treatment as described in (A–F). (H) Serum TNF levels at 1 h after treatments described in (A–F). Data are mean ± SD for 4–9 mice/group. ** p < 0.01 vs PBS, other TLR ligands and TNF-treated mice, * p < 0.05 vs PBS. Cleaved caspase 3 stained proximal small intestine sections from (I) WT and (J) TNF−/− mice at 3 h after i.p. injection of 30 µg/g pIC. Scale bar 100 µm. (K) Cleaved caspase 3 labeled cells per villus in proximal small intestine sections 3 h after 30 µg/g i.p. pIC. (L) Serum TNF levels at 1 h following i.p. injection of 30 µg/g pIC in WT and TNF−/− mice. Values are mean ± SD, n=9–14 mice/group.
pIC-induced IEC apoptosis is independent of TNF, IL-6 and IL-1 signaling
We definitively excluded the involvement of TNF in pIC-induced apoptosis using mice deficient in TNF (TNF−/−) that did not produce TNF following dsRNA treatment (Fig. 3 L). TNF−/− mice were as sensitive as WT mice to dsRNA-induced IEC apoptosis (Fig. 3 I–K). These results, confirmed that TNF has no role in pIC-induced small intestinal IEC apoptosis. We also confirmed that IL-6 and IL-1β, two cytokines that were increased after pIC, are not required for dsRNA-induced IEC apoptosis using IL-6−/− and IL-1R−/− mice (Fig. S3).
IEC apoptosis is independent of IL-15 and NK cells
IL-15 signaling and NK cell mediated killing were reported to be required for dsRNA-induced small intestinal villus shortening and epithelial cell injury (4). To evaluate the role of IL-15 in our model, we utilized mice deficient in IL-15 production. IL-15−/− mice demonstrated the same level of sensitivity to pIC as WT mice (Fig. 4 A–D). To assess the role of NK1.1+ cells in dsRNA-induced IEC apoptosis, those cells were depleted using antibody to NK1.1. We observed the same level of IEC apoptosis and small intestinal damage in NK1.1 depleted mice as in littermates receiving control IgG (Fig. 4 E–H). NK1.1+ cell depletion was confirmed by immunohistochemistry (Fig. S4 A–C).
Figure 4. IL-15 signaling, NK+ cells and type I IFN signaling are not required for pIC-induced villus shortening and IEC apoptosis.
Cleaved caspase 3 stained proximal small intestine sections from (A) WT and (B) IL-15−/− mice 3 h after 30 µg/g i.p. pIC. (C) Proximal small intestine villus length and (D) cleaved caspase 3 labeled cells per villus following the treatments in (A) and (B) at 3 h. Cleaved caspase 3 stained proximal small intestine sections for WT mice treated with (E) antibody isotype control IgG2a or (F) NK1.1 antibody for 24 h prior to 3 h 30 µg/g i.p. pIC treatment. (G) Proximal small intestine villus length and (H) cleaved caspase 3 labeled cells per villus for mice treated as in (E) and (F). Data are mean ± SD, n=5–9 mice/group. (I) IFN-β mRNA levels normalized to GAPDH using quantitative real time PCR analysis of WT and TLR3−/− mice treated with PBS or 30 µg/g i.p. pIC. RT used RNA extracted from samples 3 h post treatment. Data are mean ± SD of normalized data, n=3–4 mice/group. ** p < 0.01 vs WT PBS and TLR3−/− pIC. Cleaved caspase 3 stained proximal small intestine sections from (J) WT, (K) IFNAR−/−, (N) WT, (O) IRF3−/− and (P) BAX−/− mice 3 h after 30 µg/g i.p. pIC. (L and Q) Proximal small intestine villus length and (M and R) cleaved caspase 3 labeled cells per villus from these pIC-treated mice at 3 h. Graphs show mean ± SD, n=4–5 mice/group. Scale bar 100 µm.
IEC apoptosis does not require type I IFN signaling or IRF3
IFN-β expression in the small intestinal mucosa was increased in WT but not TLR3−/− mice after pIC (Fig. 4 I). Because type I IFN signaling was reported to potentiate virus-induced apoptosis (36), we assessed the involvement of type I IFN signaling in pIC-induced IEC apoptosis and villus shortening. There was no significant difference between IFNAR−/− and WT mice in villus shortening or apoptosis (Fig. 4 J–M).
Antiviral signaling through MAVS can activate IRF3 in a manner that results in IRF3 interacting with BAX and localizing to mitochondria to induce apoptosis (6). Because TLR3 signaling also activates IRF3 (37), we tested if this pathway was involved in pIC activated IEC apoptosis. We found no evidence to suggest the involvement of IRF3 or BAX, since neither IRF3−/− nor BAX−/− mice manifested differences in villus length or IEC apoptosis compared to WT mice after pIC treatment (Fig. 4 N–R).
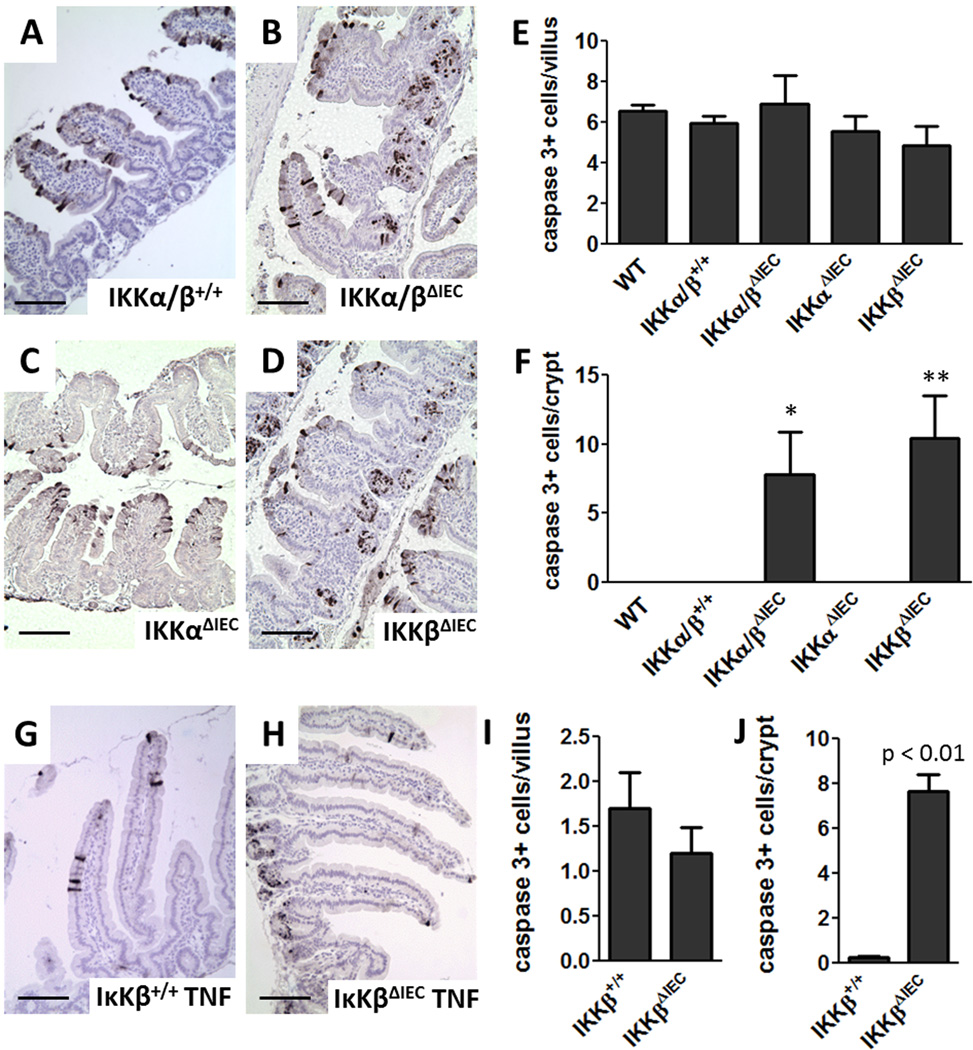
NF-κB signaling in IECs does not affect villus IEC apoptosis, but is required to protect IECs in the small intestinal crypts from apoptosis
TLR3-TRIF signaling can activate the transcription factor NF-κB (37). To assess the involvement of NF-κB signaling in pIC induced apoptosis of IECs, we used mice conditionally deficient in IKKα and β in small intestinal IECs (27, 38). The magnitude of IEC apoptosis in the villi of IKKα/βΔIEC and control mice was similar after pIC (Fig. 5 B and E), whereas IKKα/βΔIEC mice had increased levels of IEC apoptosis in the small intestinal crypts (Fig. 5 B, F). To assess the contribution of IKKα and IKKβ to this phenotype, we used IKKαΔIEC and IKKβΔIEC mice deficient selectively in IKKα or IKKβ, respectively, in the IECs and determined that increased crypt apoptosis after pIC was dependent on the absence of IKKβ signaling (Fig. 5 D and F). Notably, this same pattern of IKKβ-dependence for crypt, but not IEC, apoptosis was observed in response to TNF (Fig. 5 G–J).
Figure 5. pIC- and TNF-induced IEC apoptosis is independent of NF-κB signaling, but NF-κB signaling protects crypt cells from apoptosis.
Cleaved caspase 3 stained proximal small intestine sections from (A) IKKα/β+/+ control, (B) IKKα/βΔIEC, (C) IKKαΔIEC and (D) IKKβΔIEC mice at 3 h after 30 µg/g i.p. pIC. Cleaved caspase 3 labeled cells 3 h after 30 µg/g i.p. pIC treatment (E) per villus and (F) per crypt. Data are mean ± SD, n=3–5 mice/group. * p < 0.05 or ** p < 0.01 compared to WT, IKKα/β+/+ and IKKαΔIEC. Cleaved caspase 3 stained proximal small intestine sections from (G) IKKβ+/+ and (H) IKKβΔIEC mice 3 h after 5 µg i.p. TNF. Cleaved caspase 3 labeled cells (I) per villus or (J) per crypt for IKKβ+/+ and IKKβΔIEC mice 3 h after 5 µg i.p. TNF. Values are mean ± SD, n=3–5 mice/group. Scale bar 100 µm.
TLR3 signaling in cells of non-hematopoietic origin is required for IEC apoptosis
NK1.1 cell depletion demonstrated that NK1.1+ cells were not involved in pIC-activated IEC apoptosis (Fig. 4 E–H). To exclude the contribution of other immune cell subsets, we assessed the sensitivity of immunodeficient RAG1−/− mice that lack functional B and T cells and certain intraepithelial lymphocyte (IEL) subsets (39) to dsRNA-induced IEC apoptosis. RAG1−/− mice were not protected from pIC-induced villus shortening and IEC apoptosis (Fig. S4 D–F). We also assessed the sensitivity of DC-depleted mice (26) to pIC-activated IEC apoptosis. CD11c-DTR mice treated with diphtheria toxin (DTX) to deplete DCs showed similar levels of IEC apoptosis as WT mice in response to pIC (Fig. S4 K–O). Interestingly, DC-depleted mice had significantly decreased serum TNF levels after pIC treatment (Fig. S4 P). DC depletion was confirmed by immunohistochemistry (Fig. S4 Q, R).
To determine if other cells of hematopoietic origin contribute to dsRNA-induced IEC apoptosis after pIC, we generated TLR3−/− bone marrow chimera mice. As shown in figure 6, WT mice reconstituted with TLR3-deficient bone marrow (TLR3−/− BM→ WT) were as sensitive to pIC-induced IEC apoptosis as WT mice (Fig 6 A and C). Additionally, TLR3−/− mice reconstituted with WT bone marrow (WT BM→TLR3−/−) did not show increased IEC apoptosis in response to pIC (Fig. 6 B and D). Numbers of cleaved caspase 3 positive IECs per villus in these mice demonstrated that IEC apoptosis was not dependent on hematopoietic cells of WT origin (Fig. 6 E). Successful bone marrow transfer was apparent from assaying serum TNF levels in mice after treatment with pIC (Fig 6 F). WT mice showed TNF levels consistent with previous experiments and TLR3−/− mice showed no induction of TNF after pIC, whereas TLR3−/− BM→WT mice had levels of TNF comparable to TLR3−/− mice and WT BM→TLR3−/− mice had TNF levels comparable to WT mice (Fig. 6 F).
Figure 6. Cells of hematopoietic origin are not required for pIC-induced IEC apoptosis.
Cleaved caspase 3 staining of (A) WT, (B) TLR3−/−, (C) TLR3−/− bone marrow reconstituted WT (TLR3−/− BM→WT), and (D) WT bone marrow reconstituted TLR3−/− (WT BM→TLR3−/−) mice following 30 µg/g pIC treatment for 3 h. Scale bar 100 µm. (E) Cleaved caspase 3 positive cells per villus and (F) TNF cytokine levels for each group. Graph show results from each mouse. Bar is mean value. *P < 0.01 compared to WT and TLR3−/− BM→WT. # P < 0.01 compared to WT and WT BM→TLR3−/−.
Caspase 8 coordinates IEC apoptosis and preserves the integrity of the intestinal epithelium following dsRNA treatment
We observed activated caspase 8 at early time points following pIC treatment (Fig. S1 D, E). TRIF has been shown to activate apoptosis through caspase 8, the same initiator caspase that mediates TNF-induced apoptosis (8, 10). To assess the importance of caspase 8 in pIC-activated IEC apoptosis and subsequent small intestinal damage, we generated mice that lack caspase 8 in IECs. Control mice with floxed caspase 8 (casp8fl) behaved like WT mice following pIC treatment and had numerous mid to upper villus IECs that were positive for activated caspase 3 (Fig. 7 B, C). However, mice lacking caspase 8 in IECs (casp8ΔIEC) did not have significant levels of activated caspase 3 in villus IECs indicative of epithelial apoptosis (Fig 7 E). Interestingly, by 2.5 h post treatment, these mice manifested destruction of the small intestinal villi, with a loss of the villus epithelium (Fig. 7 F), and died within 6 h of treatment (data not shown). These results demonstrate that caspase 8 is required to regulate IEC apoptosis following pIC activated TLR3-TRIF signaling in villus IECs. In the absence of caspase 8, signaling through TLR3-TRIF causes villus destruction and death.
Figure 7. Caspase 8 is required for IEC apoptosis and preservation of the epithelium after pIC treatment.

Cleaved caspase 3 stained proximal small intestine sections from Caspase 8fl control mice treated with (A) PBS or 30 µg/g i.p pIC for (B) 1.5 h or (C) 2.5 h and mice lacking caspase 8 in their IECs (casp8ΔIEC) treated with (D) PBS or 30 µg/g i.p pIC for (E) 1.5 h or (F) 2.5 h. Scale bar 100 µm.
DISCUSSION
We elucidated the signaling pathways and mechanisms by which the viral dsRNA mimic pIC selectively causes small intestinal mucosal damage in vivo. Small intestinal mucosal changes after encountering pIC were characterized by a rapid onset of epithelial cell apoptosis, the loss of IECs from villus structures, accompanying marked loss of villus height, and significant diarrhea with recovery over the following 24–48 h. IEC apoptosis and villus shortening after pIC treatment were strictly dependent on signaling through the TLR3-TRIF pathway and caspase 8 as demonstrated using TLR3−/−, TRIF−/−, and caspase 8ΔIEC mice. While this apoptotic pathway has been studied in vitro (40), we demonstrate an in vivo system where it dramatically impacts the structure and function of the small intestine.
TNF is also well known for its ability to induce IEC apoptosis (12), but dsRNA-induced IEC apoptosis was completely independent of TNF as well as other cytokines like IL-6 and IL-1. Moreover, pIC-induced apoptosis of small intestinal villus IECs in vivo was independent of the TRIF signaling arm of TLR4 and dsRNA signaling pathways that use the MAVS adaptor molecule or PKR.
Induction of type I IFN in IECs is an important antiviral mechanism (41, 42). However, pIC-induced IEC apoptosis in the villi and villus shortening was independent of this pathway. Further, while IRF3 can induce apoptosis in response to viral infection through a BAX-dependent pathway in fibroblast cell lines (6), we found no role for BAX and IRF3 in pIC-induced apoptosis of small intestinal IECs.
dsRNA-induced small intestinal epithelial cell apoptosis, mucosal remodeling and recovery after pIC in vivo required signaling downstream of TLR3-TRIF through caspase 8. In the absence of an intact signaling pathway through caspase 8 there was severe damage to the epithelium and small intestinal mucosa. This suggests the caspase 8 apoptotic pathway has an important function as a protective mechanism that maintains the integrity of the intestinal epithelium after dsRNA-activated TLR3-TRIF signaling. Moreover, the lack of significant caspase 3 activation in pIC treated villus IECs from caspase 8ΔIEC mice suggests IEC death and loss by an alternative death pathway such as necroptosis, as seen after TNF treatment (16, 40, 43).
Cells generated in the stem cell compartment in the crypts of the small intestine proliferate and migrate upwards to populate the villi where they differentiate and ultimately are shed from the upper villus tips. Apoptosis of IECs after pIC was largely confined to the mid to upper villus region. Activation of NF-κB in IECs is important both for the induction of IEC innate proinflammatory chemokines and cytokines and for IEC resistance to apoptosis (27, 38, 44, 45). Using mice conditionally deficient in IKKα, IKKβ, or both in IECs, we showed that activation of the canonical NF-κB pathway protected crypt IECs but not villus IECs from pIC-induced as well as TNF-induced apoptosis. Moreover, the marked sensitivity to dsRNA-induced apoptosis of mid to upper villus IECs in the small intestine occurred in the absence of additional stimuli. This differs from many other cell types and tissues that require two stimuli to induce apoptosis (e.g., the activation of death receptors and translation inhibition) (11). For example, this is the case in vivo for hepatocytes where both TNF and D-galactosamine, a translation inhibitor, are required to induce apoptosis (46). Even in certain cancer cells, dsRNA-induced apoptosis through TLR3 requires a translation inhibitor (47). In those cells, pro-survival signals from NF-κB signaling appear to protect from the activation of apoptosis by caspase-8 (11, 48). It is possible that the different susceptibility of crypt and villus IECs to apoptosis may reflect differences in the negative regulation of NF-κB signaling in these cells, the state of proliferation or differentiation of those cells, or different NF-κB isoforms throughout the crypt villus axis, where an increased fraction of p50 homodimers may act to negatively regulate NF-κB targets in the villi (38, 49, 50). In this regard, NF-κB signaling likely is tightly regulated in villus IECs since these cells ultimately must undergo detachment-dependent apoptosis, termed anoikis, at the villus tips as a part of their lifecycle and NF-κB signaling has been shown to decrease anoikis (51).
Infection with enteric viruses like rotavirus causes significant loss of small intestinal villus epithelial cells, villus shortening and concomitant diarrhea (52, 53) similar to that noted herein. Other Reoviridae also contain a dsRNA genome whereas other viruses that induce gastroenteritis, including norovirus, produce dsRNA during their replication cycle (54–56). The likely relevance of the model using parenterally administered synthetic dsRNA to enteric virus infection is strengthened further since circulating dsRNA is found in rotavirus infected humans and animals (57–60). Our findings with dsRNA suggest a host protective role for the TLR3-TRIF-Caspase 8 activated apoptosis pathway in IECs in viral infection in vivo. This would result in a rapid shedding of apoptotic virus-infected cells with shortening of villi and a decreased epithelial surface area available for virus infection. Further, the generation of fluid in the intestinal lumen by crypt secretory epithelium concurrent with less epithelial surface available for fluid absorption would culminate in diarrhea that flushes out virus. Conversely, while TLR3−/− and TRIF−/− mice are protected from dsRNA induced epithelial cell apoptosis, villus remodeling and diarrhea, therapeutic approaches designed to inhibit TLR3 signaling could result in decreased virus clearance, as was reported in TLR3−/− mice infected with murine rotavirus (61).
The mechanisms, which we show are responsible for pIC-induced small intestinal epithelial cell death and villus shortening, differ from those proposed by Zhou et al (4, 62). Their model was based on a mix of in vitro and in vivo experiments in which pIC signaling through TLR3 was proposed to increase IL-15 production by IECs and Rae1, a ligand for NKG2D on IECs. Further, IEC produced IL-15 was proposed to increase NKG2D on IELs, with co-culture of isolated IECs and IELs resulting in killing of IECs in vitro at 24 h. Support for their model of pIC-induced small intestinal injury was based on an in vivo finding that antibody to IL-15 Rα partially decreased villus shortening in pIC-injected mice (62). Whereas dsRNA has been reported to induce IL-15 in vivo (63), using IL-15−/− mice we showed that pIC-induced apoptosis and villus shortening after pIC was totally independent of IL-15. Zhou et al (62) further reported decreased small intestinal villus damage in response to dsRNA after NK1.1 cell depletion, whereas NK1.1 cell depletion did not affect dsRNA induced IEC apoptosis or villus shortening in our studies. Additionally, using bone marrow chimera mice we showed that cells of hematopoietic origin were not required for IEC apoptosis following pIC treatment.
Finally, the relevance of our findings can be envisioned to extend beyond viral RNA triggers of TLR3-TRIF signaling. In this regard mRNA released from necrotic cells was shown to activate TLR3 signaling (64). Further, RNA from necrotic cells in rheumatoid arthritis synovial tissues activated TLR3-dependent signaling (65). In other studies, endogenous RNA from injured tissues and cells activated TLR3 signaling, and resulted in cell death. For example, endogenous RNA, derived from lung tissue or necrotic neutrophils, signaled through TLR3 in the absence of exogenous virus and increased lung damage, and death in a model of hyperoxia lung injury (66). TLR3 also functioned as an endogenous sensor of tissue necrosis, independent of viral activation, in models of septic peritonitis and ischemic gut injury (67). Importantly, injury and mortality were attenuated in anti-TLR3 antibody treated and TLR3−/− mice. Taken together with our results, these studies indicate that dsRNA associated with either virus infection or tissue injury and inflammation can have a dramatic impact on mucosal tissues.
Supplementary Material
ACKNOWLEDGEMENTS
We thank Sandra M. Peterson, Daniel T. Dempsey, Michael R. Wang and Kim N. Vu for expert technical assistance. We thank Eyal Raz for providing IL-1R−/− mice, Michael David for providing IRF3−/− mice, and Ekihiro Seki for providing TLR4−/− mice. We thank Elaine Hanson and Lucia Hall for maintenance of the mouse colony. We thank Dr. Nissi Varki for assistance with immunohistochemistry.
Abbreviations
- BAX
Bcl-2 associated X protein
- DC
dendritic cell
- dsRNA
double-stranded RNA
- IEC
intestinal epithelial cells
- IFN
interferon
- IKK
IκB kinase
- IRF
interferon regulatory factor
- MAVS
mitochondrial antiviral-signaling protein
- pIC
polyinosinic:polycytidylic acid
- PKR
double-stranded RNA-activated protein kinase
- PRR
pathogen pattern recognition receptor
- TLR
Toll-like receptor
- TNF
tumor necrosis factor
- TRIF
TIR-domain-containing adapter-inducing interferon-β
Footnotes
This work was supported by National Institutes of Health grants DK35108 and DK80506 and a grant from the William K. Warren Foundation.
The authors have no conflicting financial interests.
REFERENCES
- 1.Abreu MT. Toll-like receptor signalling in the intestinal epithelium: how bacterial recognition shapes intestinal function. Nat Rev Immunol. 2010;10:131–144. doi: 10.1038/nri2707. [DOI] [PubMed] [Google Scholar]
- 2.Kawai T, Akira S. Toll-like receptor and RIG-I-like receptor signaling. Ann N Y Acad Sci. 2008;1143:1–20. doi: 10.1196/annals.1443.020. [DOI] [PubMed] [Google Scholar]
- 3.Thompson AJV, Locarnini SA. Toll-like receptors, RIG-I-like RNA helicases and the antiviral innate immune response. Immunol Cell Biol. 2007;85:435–445. doi: 10.1038/sj.icb.7100100. [DOI] [PubMed] [Google Scholar]
- 4.Zhou R, Wei H, Sun R, Tian Z. Recognition of double-stranded RNA by TLR3 induces severe small intestinal injury in mice. J Immunol. 2007;178:4548–4556. doi: 10.4049/jimmunol.178.7.4548. [DOI] [PubMed] [Google Scholar]
- 5.Clarke P, Tyler KL. Apoptosis in animal models of virus-induced disease. Nat Rev Micro. 2009;7:144–155. doi: 10.1038/nrmicro2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chattopadhyay S, Marques JT, Yamashita M, Peters KL, Smith K, Desai A, Williams BR, Sen GC. Viral apoptosis is induced by IRF-3-mediated activation of Bax. EMBO J. 2010;29:1762–1773. doi: 10.1038/emboj.2010.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kalai M, Van Loo G, Vanden Berghe T, Meeus A, Burm W, Saelens X, Vandenabeele P. Tipping the balance between necrosis and apoptosis in human and murine cells treated with interferon and dsRNA. Cell Death Differ. 2002;9:981–994. doi: 10.1038/sj.cdd.4401051. [DOI] [PubMed] [Google Scholar]
- 8.Kaiser WJ, Offermann MK. Apoptosis induced by the toll-like receptor adaptor TRIF is dependent on its receptor interacting protein homotypic interaction motif. J Immunol. 2005;174:4942–4952. doi: 10.4049/jimmunol.174.8.4942. [DOI] [PubMed] [Google Scholar]
- 9.Feoktistova M, Geserick P, Kellert B, Dimitrova DP, Langlais C, Hupe M, Cain K, MacFarlane M, Hacker G, Leverkus M. cIAPs block Ripoptosome formation, a RIP1/caspase-8 containing intracellular cell death complex differentially regulated by cFLIP isoforms. Mol Cell. 2011;43:449–463. doi: 10.1016/j.molcel.2011.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Estornes Y, Toscano F, Virard F, Jacquemin G, Pierrot A, Vanbervliet B, Bonnin M, Lalaoui N, Mercier-Gouy P, Pacheco Y, Salaun B, Renno T, Micheau O, Lebecque S. dsRNA induces apoptosis through an atypical death complex associating TLR3 to caspase-8. Cell Death Differ. 2012 doi: 10.1038/cdd.2012.22. Epub. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Van Herreweghe F, Festjens N, Declercq W, Vandenabeele P. Tumor necrosis factor-mediated cell death: to break or to burst, that's the question. Cell Mol Life Sci. 2010;67:1567–1579. doi: 10.1007/s00018-010-0283-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Piguet PF, Vesin C, Guo J, Donati Y, Barazzone C. TNF-induced enterocyte apoptosis in mice is mediated by the TNF receptor 1 and does not require p53. Eur J Immunol. 1998;28:3499–3505. doi: 10.1002/(SICI)1521-4141(199811)28:11<3499::AID-IMMU3499>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 13.Lau KS, Juchheim AM, Cavaliere KR, Philips SR, Lauffenburger DA, Haigis KM. In vivo systems analysis identifies spatial and temporal aspects of the modulation of TNF-alpha-induced apoptosis and proliferation by MAPKs. Sci Signal. 2011;4:ra16. doi: 10.1126/scisignal.2001338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Beisner DR, Ch'en IL, Kolla RV, Hoffmann A, Hedrick SM. Cutting edge: innate immunity conferred by B cells is regulated by caspase-8. J Immunol. 2005;175:3469–3473. doi: 10.4049/jimmunol.175.6.3469. [DOI] [PubMed] [Google Scholar]
- 15.Madison BB, Dunbar L, Qiao XT, Braunstein K, Braunstein E, Gumucio DL. Cis elements of the villin gene control expression in restricted domains of the vertical (crypt) and horizontal (duodenum, cecum) axes of the intestine. J Biol Chem. 2002;277:33275–33283. doi: 10.1074/jbc.M204935200. [DOI] [PubMed] [Google Scholar]
- 16.Gunther C, Martini E, Wittkopf N, Amann K, Weigmann B, Neumann H, Waldner MJ, Hedrick SM, Tenzer S, Neurath MF, Becker C. Caspase-8 regulates TNF-alpha-induced epithelial necroptosis and terminal ileitis. Nature. 2011;477:335–339. doi: 10.1038/nature10400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hoebe K, Du X, Georgel P, Janssen E, Tabeta K, Kim SO, Goode J, Lin P, Mann N, Mudd S, Crozat K, Sovath S, Han J, Beutler B. Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature. 2003;424:743–748. doi: 10.1038/nature01889. [DOI] [PubMed] [Google Scholar]
- 18.Knudson CM, Tung KS, Tourtellotte WG, Brown GA, Korsmeyer SJ. Bax-deficient mice with lymphoid hyperplasia and male germ cell death. Science. 1995;270:96–99. doi: 10.1126/science.270.5233.96. [DOI] [PubMed] [Google Scholar]
- 19.Mombaerts P, Iacomini J, Johnson RS, Herrup K, Tonegawa S, Papaioannou VE. RAG-1-deficient mice have no mature B and T lymphocytes. Cell. 1992;68:869–877. doi: 10.1016/0092-8674(92)90030-g. [DOI] [PubMed] [Google Scholar]
- 20.Kopf M, Baumann H, Freer G, Freudenberg M, Lamers M, Kishimoto T, Zinkernagel R, Bluethmann H, Kohler G. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature. 1994;368:339–342. doi: 10.1038/368339a0. [DOI] [PubMed] [Google Scholar]
- 21.Glaccum MB, Stocking KL, Charrier K, Smith JL, Willis CR, Maliszewski C, Livingston DJ, Peschon JJ, Morrissey PJ. Phenotypic and functional characterization of mice that lack the type I receptor for IL-1. J Immunol. 1997;159:3364–3371. [PubMed] [Google Scholar]
- 22.Kennedy MK, Glaccum M, Brown SN, Butz EA, Viney JL, Embers M, Matsuki N, Charrier K, Sedger L, Willis CR, Brasel K, Morrissey PJ, Stocking K, Schuh JC, Joyce S, Peschon JJ. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J Exp Med. 2000;191:771–780. doi: 10.1084/jem.191.5.771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Van Uden JH, Tran CH, Carson DA, Raz E. Type I interferon is required to mount an adaptive response to immunostimulatory DNA. Eur J Immunol. 2001;31:3281–3290. doi: 10.1002/1521-4141(200111)31:11<3281::aid-immu3281>3.0.co;2-0. [DOI] [PubMed] [Google Scholar]
- 24.Yang YL, Reis LF, Pavlovic J, Aguzzi A, Schafer R, Kumar A, Williams BR, Aguet M, Weissmann C. Deficient signaling in mice devoid of double-stranded RNA-dependent protein kinase. EMBO J. 1995;14:6095–6106. doi: 10.1002/j.1460-2075.1995.tb00300.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Honda K, Sakaguchi S, Nakajima C, Watanabe A, Yanai H, Matsumoto M, Ohteki T, Kaisho T, Takaoka A, Akira S, Seya T, Taniguchi T. Selective contribution of IFN-alpha/beta signaling to the maturation of dendritic cells induced by double-stranded RNA or viral infection. Proc Natl Acad Sci U S A. 2003;100:10872–10877. doi: 10.1073/pnas.1934678100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Abe K, Nguyen KP, Fine SD, Mo J-H, Shen C, Shenouda S, Corr M, Jung S, Lee J, Eckmann L, Raz E. Conventional dendritic cells regulate the outcome of colonic inflammation independently of T cells. Proceedings of the National Academy of Sciences. 2007;104:17022–17027. doi: 10.1073/pnas.0708469104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Eckmann L, Nebelsiek T, Fingerle AA, Dann SM, Mages J, Lang R, Robine S, Kagnoff MF, Schmid RM, Karin M, Arkan MC, Greten FR. Opposing functions of IKKbeta during acute and chronic intestinal inflammation. Proc Natl Acad Sci U S A. 2008;105:15058–15063. doi: 10.1073/pnas.0808216105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nenci A, Becker C, Wullaert A, Gareus R, van Loo G, Danese S, Huth M, Nikolaev A, Neufert C, Madison B, Gumucio D, Neurath MF, Pasparakis M. Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature. 2007;446:557–561. doi: 10.1038/nature05698. [DOI] [PubMed] [Google Scholar]
- 29.Hoshino K, Takeuchi O, Kawai T, Sanjo H, Ogawa T, Takeda Y, Takeda K, Akira S. Cutting edge: Toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J Immunol. 1999;162:3749–3752. [PubMed] [Google Scholar]
- 30.Sato M, Suemori H, Hata N, Asagiri M, Ogasawara K, Nakao K, Nakaya T, Katsuki M, Noguchi S, Tanaka N, Taniguchi T. Distinct and essential roles of transcription factors IRF-3 and IRF-7 in response to viruses for IFN-alpha/beta gene induction. Immunity. 2000;13:539–548. doi: 10.1016/s1074-7613(00)00053-4. [DOI] [PubMed] [Google Scholar]
- 31.Sun Q, Sun L, Liu HH, Chen X, Seth RB, Forman J, Chen ZJ. The specific and essential role of MAVS in antiviral innate immune responses. Immunity. 2006;24:633–642. doi: 10.1016/j.immuni.2006.04.004. [DOI] [PubMed] [Google Scholar]
- 32.Maaser C, Housley MP, Iimura M, Smith JR, Vallance BA, Finlay BB, Schreiber JR, Varki NM, Kagnoff MF, Eckmann L. Clearance of Citrobacter rodentium requires B cells but not secretory immunoglobulin A (IgA) or IgM antibodies. Infect Immun. 2004;72:3315–3324. doi: 10.1128/IAI.72.6.3315-3324.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gareau MG, Jury J, MacQueen G, Sherman PM, Perdue MH. Probiotic treatment of rat pups normalises corticosterone release and ameliorates colonic dysfunction induced by maternal separation. Gut. 2007;56:1522–1528. doi: 10.1136/gut.2006.117176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fan Y, Bergmann A. The cleaved-Caspase-3 antibody is a marker of Caspase-9-like DRONC activity in Drosophila. Cell Death Differ. 2009;17:534–539. doi: 10.1038/cdd.2009.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Scheuner D, Patel R, Wang F, Lee K, Kumar K, Wu J, Nilsson A, Karin M, Kaufman RJ. Double-stranded RNA-dependent protein kinase phosphorylation of the alpha-subunit of eukaryotic translation initiation factor 2 mediates apoptosis. J Biol Chem. 2006;281:21458–21468. doi: 10.1074/jbc.M603784200. [DOI] [PubMed] [Google Scholar]
- 36.Balachandran S, Roberts PC, Kipperman T, Bhalla KN, Compans RW, Archer DR, Barber GN. Alpha/beta interferons potentiate virus-induced apoptosis through activation of the FADD/Caspase-8 death signaling pathway. J Virol. 2000;74:1513–1523. doi: 10.1128/jvi.74.3.1513-1523.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jiang Z, Mak TW, Sen G, Li X. Toll-like receptor 3-mediated activation of NF-kappaB and IRF3 diverges at Toll-IL-1 receptor domain-containing adapter inducing IFN-beta. Proc Natl Acad Sci U S A. 2004;101:3533–3538. doi: 10.1073/pnas.0308496101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Egan LJ, Eckmann L, Greten FR, Chae S, Li ZW, Myhre GM, Robine S, Karin M, Kagnoff MF. IkappaB-kinasebeta-dependent NF-kappaB activation provides radioprotection to the intestinal epithelium. Proc Natl Acad Sci U S A. 2004;101:2452–2457. doi: 10.1073/pnas.0306734101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Page ST, van Oers NS, Perlmutter RM, Weiss A, Pullen AM. Differential contribution of Lck and Fyn protein tyrosine kinases to intraepithelial lymphocyte development. Eur J Immunol. 1997;27:554–562. doi: 10.1002/eji.1830270229. [DOI] [PubMed] [Google Scholar]
- 40.Mocarski ES, Upton JW, Kaiser WJ. Viral infection and the evolution of caspase 8-regulated apoptotic and necrotic death pathways. Nat Rev Immunol. 2011;12:79–88. doi: 10.1038/nri3131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Broquet AH, Hirata Y, McAllister CS, Kagnoff MF. RIG-I/MDA5/MAVS are required to signal a protective IFN response in rotavirus-infected intestinal epithelium. J Immunol. 2011;186:1618–1626. doi: 10.4049/jimmunol.1002862. [DOI] [PubMed] [Google Scholar]
- 42.Hirata Y, Broquet AH, Menchen L, Kagnoff MF. Activation of innate immune defense mechanisms by signaling through RIG-I/IPS-1 in intestinal epithelial cells. J Immunol. 2007;179:5425–5432. doi: 10.4049/jimmunol.179.8.5425. [DOI] [PubMed] [Google Scholar]
- 43.Linkermann A, Brasen JH, De Zen F, Weinlich R, Schwendener RA, Green DR, Kunzendorf U, Krautwald S. Dichotomy between RIP1- and RIP3-mediated Necroptosis in Tumor Necrosis Factor alpha-induced Shock. Mol Med. 2012 doi: 10.2119/molmed.2011.00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Elewaut D, DiDonato JA, Kim JM, Truong F, Eckmann L, Kagnoff MF. NF-kappa B is a central regulator of the intestinal epithelial cell innate immune response induced by infection with enteroinvasive bacteria. J Immunol. 1999;163:1457–1466. [PubMed] [Google Scholar]
- 45.Chae S, Eckmann L, Miyamoto Y, Pothoulakis C, Karin M, Kagnoff MF. Epithelial cell I kappa B-kinase beta has an important protective role in Clostridium difficile toxin A-induced mucosal injury. J Immunol. 2006;177:1214–1220. doi: 10.4049/jimmunol.177.2.1214. [DOI] [PubMed] [Google Scholar]
- 46.Leist M, Gantner F, Bohlinger I, Tiegs G, Germann PG, Wendel A. Tumor necrosis factor-induced hepatocyte apoptosis precedes liver failure in experimental murine shock models. Am J Pathol. 1995;146:1220–1234. [PMC free article] [PubMed] [Google Scholar]
- 47.Jiang Q, Wei H, Tian Z. Poly I:C enhances cycloheximide-induced apoptosis of tumor cells through TLR3 pathway. BMC Cancer. 2008;8:12. doi: 10.1186/1471-2407-8-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Plumpe J, Malek NP, Bock CT, Rakemann T, Manns MP, Trautwein C. NF-kappaB determines between apoptosis and proliferation in hepatocytes during liver regeneration. Am J Physiol Gastrointest Liver Physiol. 2000;278:G173–G183. doi: 10.1152/ajpgi.2000.278.1.G173. [DOI] [PubMed] [Google Scholar]
- 49.Inan MS, Tolmacheva V, Wang QS, Rosenberg DW, Giardina C. Transcription factor NF-kappaB participates in regulation of epithelial cell turnover in the colon. Am J Physiol Gastrointest Liver Physiol. 2000;279:G1282–G1291. doi: 10.1152/ajpgi.2000.279.6.G1282. [DOI] [PubMed] [Google Scholar]
- 50.Tong X, Yin L, Washington R, Rosenberg DW, Giardina C. The p50-p50 NF-kappaB complex as a stimulus-specific repressor of gene activation. Mol Cell Biochem. 2004;265:171–183. doi: 10.1023/b:mcbi.0000044394.66951.4d. [DOI] [PubMed] [Google Scholar]
- 51.Toruner M, Fernandez-Zapico M, Sha JJ, Pham L, Urrutia R, Egan LJ. Antianoikis effect of nuclear factor-kappaB through up-regulated expression of osteoprotegerin, BCL-2, and IAP-1. J Biol Chem. 2006;281:8686–8696. doi: 10.1074/jbc.M512178200. [DOI] [PubMed] [Google Scholar]
- 52.Davidson GP, Barnes GL. Structural and functional abnormalities of the small intestine in infants and young children with rotavirus enteritis. Acta Paediatr Scand. 1979;68:181–186. doi: 10.1111/j.1651-2227.1979.tb04986.x. [DOI] [PubMed] [Google Scholar]
- 53.Salim AF, Phillips AD, Walker-Smith JA, Farthing MJ. Sequential changes in small intestinal structure and function during rotavirus infection in neonatal rats. Gut. 1995;36:231–238. doi: 10.1136/gut.36.2.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Otto PH, Clarke IN, Lambden PR, Salim O, Reetz J, Liebler-Tenorio EM. Infection of calves with bovine norovirus GIII.1 strain Jena virus: an experimental model to study the pathogenesis of norovirus infection. Journal of Virology. 2011;85:12013–12021. doi: 10.1128/JVI.05342-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ahlquist P. Parallels among positive-strand RNA viruses, reverse-transcribing viruses and double-stranded RNA viruses. Nat Rev Microbiol. 2006;4:371–382. doi: 10.1038/nrmicro1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.McCartney SA, Thackray LB, Gitlin L, Gilfillan S, Virgin HW, Colonna M. MDA-5 recognition of a murine norovirus. PLoS Pathog. 2008;4:e1000108. doi: 10.1371/journal.ppat.1000108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Blutt SE, Fenaux M, Warfield KL, Greenberg HB, Conner ME. Active viremia in rotavirus-infected mice. J. Virol. 2006;80:6702–6705. doi: 10.1128/JVI.00329-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Chiappini E, Azzari C, Moriondo M, Galli L, de Martino M. Viraemia is a common finding in immunocompetent children with rotavirus infection. J. Med. Virol. 2005;76:265–267. doi: 10.1002/jmv.20351. [DOI] [PubMed] [Google Scholar]
- 59.Crawford SE, Patel DG, Cheng E, Berkova Z, Hyser JM, Ciarlet M, Finegold MJ, Conner ME, Estes MK. Rotavirus viremia and extraintestinal viral infection in the neonatal rat model. J. Virol. 2006;80:4820–4832. doi: 10.1128/JVI.80.10.4820-4832.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Moon S, Wang Y, Dennehy P, Simonsen KA, Zhang J, Jiang B. Antigenemia, RNAemia, and innate immunity in children with acute rotavirus diarrhea. FEMS Immunol. Med. Microbiol. 2012;64:382–391. doi: 10.1111/j.1574-695X.2011.00923.x. [DOI] [PubMed] [Google Scholar]
- 61.Pott J, Stockinger S, Torow N, Smoczek A, Lindner C, McInerney G, Backhed F, Baumann U, Pabst O, Bleich A, Hornef MW. Age-Dependent TLR3 Expression of the Intestinal Epithelium Contributes to Rotavirus Susceptibility. PLoS Pathog. 2012;8:e1002670. doi: 10.1371/journal.ppat.1002670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zhou R, Wei H, Sun R, Zhang J, Tian Z. NKG2D recognition mediates Toll-like receptor 3 signaling-induced breakdown of epithelial homeostasis in the small intestines of mice. Proc Natl Acad Sci U S A. 2007;104:7512–7515. doi: 10.1073/pnas.0700822104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lodolce JP, Burkett PR, Boone DL, Chien M, Ma A. T cell-independent interleukin 15Ralpha signals are required for bystander proliferation. J Exp Med. 2001;194:1187–1194. doi: 10.1084/jem.194.8.1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kariko K, Ni H, Capodici J, Lamphier M, Weissman D. mRNA is an endogenous ligand for Toll-like receptor 3. J Biol Chem. 2004;279:12542–12550. doi: 10.1074/jbc.M310175200. [DOI] [PubMed] [Google Scholar]
- 65.Brentano F, Schorr O, Gay RE, Gay S, Kyburz D. RNA released from necrotic synovial fluid cells activates rheumatoid arthritis synovial fibroblasts via Toll-like receptor 3. Arthritis Rheum. 2005;52:2656–2665. doi: 10.1002/art.21273. [DOI] [PubMed] [Google Scholar]
- 66.Murray LA, Knight DA, McAlonan L, Argentieri R, Joshi A, Shaheen F, Cunningham M, Alexopolou L, Flavell RA, Sarisky RT, Hogaboam CM. Deleterious role of TLR3 during hyperoxia-induced acute lung injury. Am J Respir Crit Care Med. 2008;178:1227–1237. doi: 10.1164/rccm.200807-1020OC. [DOI] [PubMed] [Google Scholar]
- 67.Cavassani KA, Ishii M, Wen H, Schaller MA, Lincoln PM, Lukacs NW, Hogaboam CM, Kunkel SL. TLR3 is an endogenous sensor of tissue necrosis during acute inflammatory events. J Exp Med. 2008;205:2609–2621. doi: 10.1084/jem.20081370. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.