Abstract
Alcoholic liver disease (ALD) is a lifestyle disease with its pathogenesis and individual predisposition governed by gene–environment interactions. Based on the “second hit” or “multiple hits” hypothesis, patients are predisposed to progressive ALD when a magic combination of gene and environmental interactions exists. Reproduction of second or multiple hits in animal models serves to test a combination and to gain mechanistic insights into synergism achieved by such combination. Numerous environmental factors have been incorporated into animal models, largely classified into nutritional, xenobiotic/pharmacologic, hemodynamic, and viral groups. A loss or gain of function genetic model has become a popular experimental approach to test the role of a gene as a second hit. Future research will need to test more subtle or natural hits combined with excessive alcohol intake to test multiple hits in the genesis of ALD. Additionally, animal models of comorbidities are urgently needed particularly for synergistic liver disease and oncogenesis caused by alcohol, obesity, and hepatitis virus.
Keywords: Gene-environment interactions, alcoholic liver disease, alcoholic steatohepatitis, nonalcoholic steatohepatitis, nonalcoholic fatty liver disease, animal models
THE SECOND HIT HYPOTHESIS
Alcoholic and nonalcoholic steatohepatitis (ASH and NASH) are the two most common lifestyle liver diseases worldwide caused by excessive intake of alcohol and calories, respectively. Pathological evolution of ASH and NASH are very similar if not identical. A majority of alcoholic or obese patients develop hepatic steatosis, but progression to steatohepatitis occurs only in 15 to 20% of the patients.1,2 Although ASH and NASH are considered as critical turning points for the increased propensity for progression to cirrhosis, a fraction (20 to 50%) of ASH and NASH patients develop cirrhosis.1,2 The fact that most alcoholic or obese patients develop fatty liver, but experience a sharp decline in the incidence of ASH/NASH and cirrhosis, has led to a proposal that excessive intake of alcohol or calories is the first hit to induce hepatic steatosis, the early stage of alcoholic liver disease (ALD), or nonalcoholic fatty liver disease (NAFLD). The second hit is required for the development of ASH or NASH and of cirrhosis (Fig. 1). The obvious and important question is what this second hit is. Indeed, this is the question addressed by many investigators, which has resulted in the generation of numerous second hit ASH or NASH animal models summarized below. However, it is very important to recognize that the second hit question cannot be discussed without understanding the gene–environment interactions that govern the pathogenesis of most chronic diseases of various organs including the liver. Most likely, distinct groups of genes participate in the pathogenesis of different stages of the ASH/NASH pathologic spectrum, and environmental factors interplay with suspected genes for their expression and participation in the pathogenesis (Fig. 2). There also exist cross-interactions within the genes as well as within environmental factors. These complex interactions between and among genetic and environmental factors must determine the difference in individual predisposition to progression of ASH and NASH. From this view, the second hit hypothesis may be too simplistic, and the insults may most likely be “multiple” (Fig. 1). Indeed, the more risk factors an individual has, the more predisposed she or he would be for progressive ASH and NASH. Thus, for the development of ASH or NASH animal models, both genetic and environmental factors need to be considered, and those which achieve most synergistic interactions are likely most successful in reproducing progressive pathology. Once such synergism is reproduced, the animal model serves to help identify its molecular mechanisms and new therapeutic targets for the disease.
Figure 1.
The “second hit” hypothesis of alcoholic steatohepatitis (ASH) and nonalcoholic steatohepatitis (NASH) pathogenesis. The second hit is most likely to be multiple hits in progression of ASH and NASH from hepatic steatosis.
Figure 2.
Gene–environment interactions that determine the individual susceptibility to alcoholic liver disease.
SECOND HIT ANIMAL MODELS FOR ALD
Table 1 summarizes a partial list of second hit animal models for alcoholic liver injury. As one may quickly realize, the listed factors and genes are known players or regulators in the pathogenesis of ALD. How some of these second hits mechanistically fit into the known mechanisms of the ALD pathogenesis is depicted by shaded boxes and bold arrows in Fig. 3. Second hit models are categorically divided into nutritional, ago-nistic/xenobiotic/pharmacologic, hemodynamic, hepatitis C virus (HCV) and genetic groups in Table 1, and are briefly reviewed with scientific backgrounds below.
Table 1.
“Second Hit” Alcoholic Liver Disease Animal Models
| Reference | |
|---|---|
| Nutritional | |
| High fat diet | 4,80 |
| Fish oil | 12,13 |
| Lron | 19,22 |
| Folate deficiency | 26 |
| Choline deficiency | 27 |
| Agonistic/Xenobiotic/Pharmacologic | |
| TLR ligands (e.g., LPS) | 36,38,81 |
| Concavalin A | 39 |
| Aldehyde adducts (acetaldehyde, MAA) | 40–43 |
| Carbon tetrachloride | 45,46 |
| Pyrazole | 44 |
| Acetaminophen | 49 |
| Epinephrine | 50 |
| Thyroid hormone | 51 |
| Hemodynamic | |
| Ischemia/reperfusion | 57 |
| Hypoxia | 58 |
| HCV | |
| Core | 64 |
| NS5A | 65 |
| Genetic | |
| Female sex | 71 |
| CYP2E1 Tg | 66 |
| Nrf2−/− | 67 |
| CuZnSOD−/− | 68 |
| IL-6−/− | 69 |
| hepSTAT3−/− | 79 |
| fa/fa | 7 |
| CBS+/− | 70 |
TLR, toll-like receptor; LPS, lipopolysaccharide; MAA, malondialdehyde-acetaldehyde adducts; CYP2E1, cytochrome P450 2E1; CuZn-SOD, copper zinc superoxide dismutase; hepSTAT3, hepatocyte specific knockout of signal transducer and activator of transcription 3; CBS, cystathionine β synthase.
Figure 3.
How diverse “second hits” interplay with known pathogenetic mechanisms of alcoholic liver disease. Shaded boxes and bold arrows depict some of the second hits discussed in this review.
Nutritional Second Hit
The most common environmental second hit approach is nutritional modification. High fat diet (HFD) has long been known to promote alcoholic liver injury in rodents. Among many effects possibly caused by HFD, the most notable is induction of cytochrome P450 2E1 (CYP2E1).3 As isocaloric feeding is usually employed, a high level of fat results in a reciprocally lower carbohydrate content, and this low carbohydrate-HFD induces CYP2E1, and consequent oxidant stress, and alcoholic liver damage.4–6 HFD also causes obesity, insulin resistance (IR), and NAFLD. Thus, this second hit may also reflect synergistic hepatic injury associated with dual effects of alcohol and obesity/IR. Further, this synergism may also be linked to CYP2E1 as over-expression of this cytochrome p450 isozyme induces hepatic IR.8 Another potential mechanism induced by HFD is endoplasmic reticulum (ER) stress also described as an unfolded protein response.9,10 ER stress is implicated in ALD,11 and HFD may worsen this condition via its ability to promote ER stress, which, in turn, induces IR.9 The type of fat is also an important consideration, and polyunsaturated fat is implicated in many of the aforementioned HFD effects. Fish oil (n-3 polyunsaturated fatty acids), in particular, is shown to aggravate experimental ALD.12,13 However, fish oil activates peroxisome proliferator–activated receptor alpha (PPARα) and suppresses de novo lipogenesis in the mouse model of steatohepatitis.14 Pretreatment with fish oil even attenuates fatty liver induced by an acute dose of ethanol, the effect which is associated with PPARα activation and reduced sterol regulatory element binding protein-1c (SREBP-1c) expression.15 These contradictory effects may be attributable to the fact that fish oil increases hepatic levels of lipid peroxides14; this effect in the setting of chronic alcohol intake may override the beneficial effects and exacerbates experimental ALD.
Iron as a Second Hit
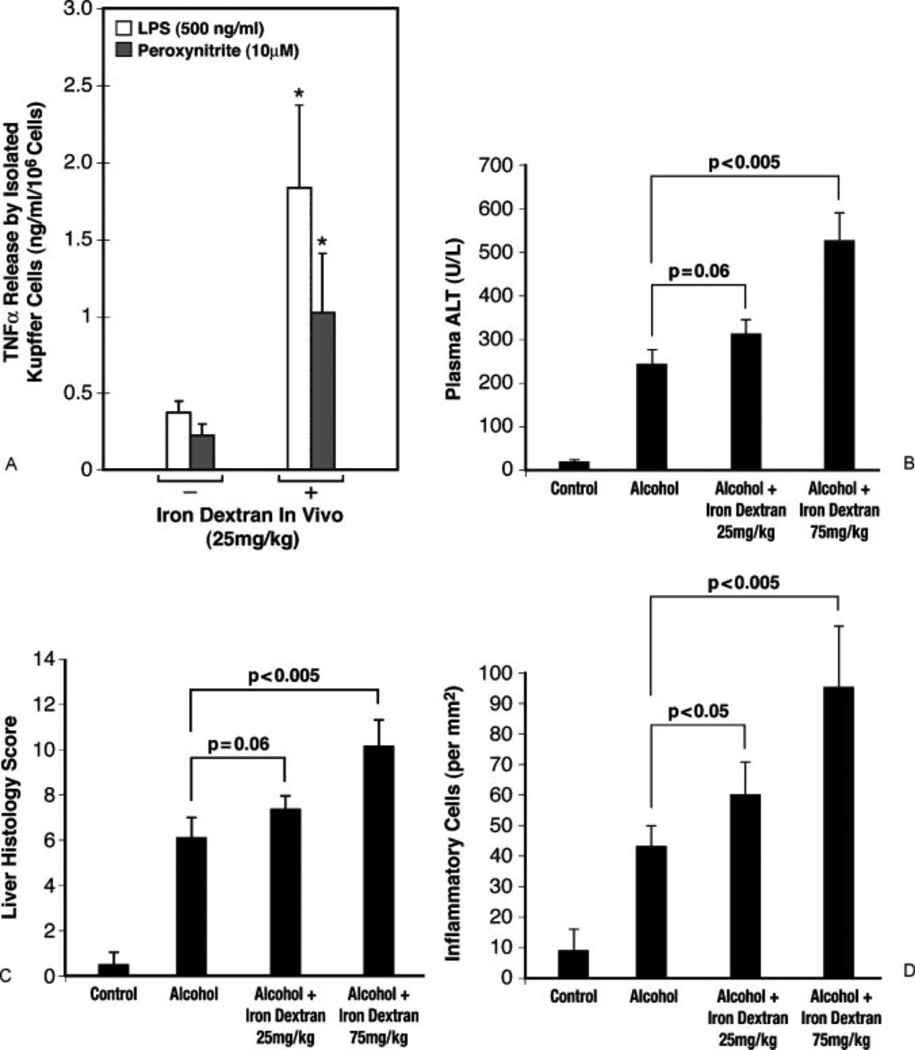
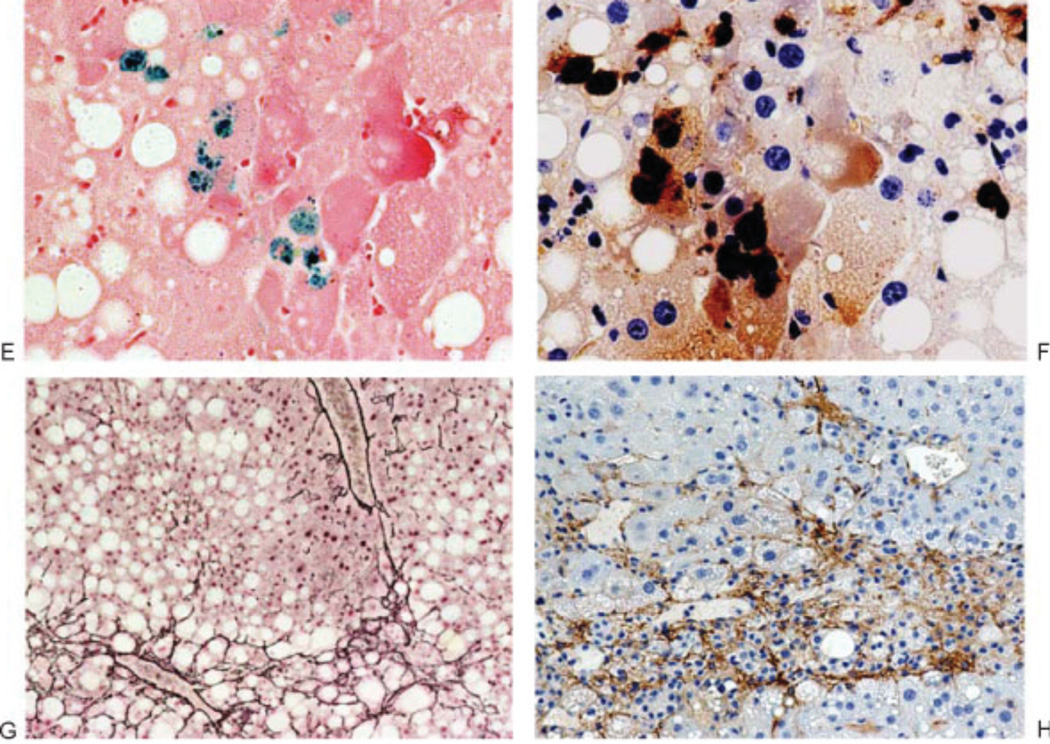
Interactions between iron and ALD have long been suggested for chronic liver disease including ALD.16 A consensus has been that iron accumulates in chronic liver inflammation and, in turn, catalyzes hydroxyl radical-mediated oxidant damage via the Fenton pathway. In fact, the treatment with an iron chelator attenuates alcoholic17 and cholestatic18 liver injury in animal models. Conversely, iron supplementation with carbonyl iron, accelerates alcoholic liver damage,19 and this iron loading can be an important second hit. The carbonyl iron method primarily achieves iron loading in hepatocytes where iron catalyzes and promotes oxidant stress and damage. However, iron also accumulates in Kupffer cells in ALD20 and NASH21 models. This iron accumulation leads to an expansion of a chelatable pool of iron, which, in turn, augments lipopolysaccharide-(LPS-) or peroxynitrite-induced iron signaling in Kupffer cells to accentuate IkappaB kinase (IKK) and nuclear factor kappa B (NFκB) activation.20,22 This novel iron signaling mechanism can be exploited to achieve a second hit. Iron dextran, if injected subcutaneously, is gradually and selectively taken up by macrophages, and this should theoretically increase iron content in macrophages including Kupffer cells and promote proinflammatory NFκB activation. Indeed, this actually happens. Kupffer cells, isolated from a normal mouse given a single dose of iron dextran, release 4- to 5-fold more tumor necrosis factor-α (TNFα) in response to LPS or peroxynitrite (Fig. 4A). This treatment in the mouse intragastric ethanol infusion model markedly exacerbates alcoholic liver injury (Fig. 4B – D). Immunostaining demonstrates localization of active p65 in hepatic macrophages (Fig. 4F) loaded with iron (Fig. 4E). This treatment even promotes liver fibro-genesis within only 4 weeks of alcohol feeding as depicted by reticulin staining (Fig. 4G) and α-smooth muscle actin staining, which identifies activated hepatic stellate cells (Fig. 4H).
Figure 4.
(A) Kupffer cells (KC) were isolated from mice at 2 weeks after a single subcutaneous injection of iron dextran (25 mg/kg) to assess lipopolysaccharide (LPS) or peroxynitrite stimulated tumor necrosis factor-α (TNFα) expression. Note KC from iron dextran-injected mice release 4 to 5 times more TNFα in response to LPS or peroxynitrite. p < 0.05 compared with the cells from mice without iron dextran injection. (B) Plasma alanine aminotransferase (ALT) levels of mice fed intragastrically with alcohol and high fat diet or pair-fed with isocaloric diet for 4 weeks without or with prior iron dextran injection. Note a single subcutaneous injection of iron dextran (75 mg/kg) prior to alcohol feeding significantly aggravates plasma ALT elevation as compared with alcohol-fed mice injected with vehicle (dextran). (C) Liver histological score of mice fed alcohol for 4 weeks without or with iron dextran injection. Note iron dextran injection at 75 mg/kg significantly worsens the liver histologic score compared with alcohol-fed mice with vehicle treatment. (D) Morphometric analysis of inflammatory cells in the liver of the different experimental groups. Note iron dextran injection significantly increases inflammatory cells in the liver as compared with alcohol-fed mice with vehicle treatment. (E) Representative microphotographs of aggravated alcoholic liver injury in iron dextran-injected mice. In the liver of alcohol-fed mice given 75 mg/kg iron dextran, hepatic macrophages stained for iron (Prussian blue reaction) are noted. (F) These iron staining-positive macrophages are also stained positively for active p65. (G) Pericellular liver fibrosis is evident in the livers of alcohol-fed mice with iron dextran treatment as demonstrated by reticulin staining. (H) Numerous activated hepatic stellate cells are present in a focus of liver necrosis and inflammation as shown by α-smooth muscle actin staining. (From Xiong S, She H, Zhang AS, et al. Hepatic macrophage iron aggravates experimental alcoholic steatohepatitis. Am J Physiol Gastrointest Liver Physiol 2008;295(3):G512–G521. Reprinted with permission from The American Physiological Society.)
Other Nutritional Second Hit Models
Chronic alcohol intake causes the deficiency of micro-nutrients such as zinc,23 folate,24 and choline,25 to list a few. Combination of alcohol intake and a diet deficient in folate26 or choline27 aggravates experimental ALD, supporting the pathogenetic importance of the micronutrient deficiencies. These deficiencies converge, besides their other independent physiological complications, to abnormal methionine metabolism well documented in ALD.28,29 Folate is required along with vitamin B12 for methionine synthesis from homocysteine catalyzed by methionine synthase. Choline constitutes a polar head for phosphatidylcholine generated by methylation of phosphatidylethanolamine using S-adenosylmethionine (SAMe) as the methyl donor, the process likely suppressed by reduced SAMe in ALD. Choline is also a precursor for trimethylglycine (betaine), which is required for another methionine resynthesis pathway via betaine-homocysteine methyltransferase (BHMT). Betaine supplementation or BHMT expression attenuates homocystinemia, SAMe depletion, and experimental ALD,30–32 supporting the role of abnormal methionine metabolism in the ALD pathogenesis. A defect in any of the key components of methionine metabolism, may serve as a second hit for ALD. Such an example is loss of function of glycine N-methyltransferase (GNMT), which results in hepatic steatosis, fibrosis and cancer in mice.33 GNMT catalyzes methylation of glycine to form N-methylglycine and by doing so removes excess SAMe and maintains a physiologic ratio of SAMe to S-adeno-sylhomocysteine (SAH) for normal methylation status. GNMT is downregulated in patients with alcoholic or/and HCV-induced liver cirrhosis,34 and as much as 40% of hepatocellular carcinoma (HCC) patients show loss of heterozygosity for GNMT.35 A study with GNMT knockout mice reveals reduced hepatic expression of the Janus kinases (Jak)/signal transducers and activators of transcription (STAT) inhibitors suppressor of cyto-kine signaling (SOCS) 1–3 and the Ras inhibitors RASSF as potential mechanistic insights into enhanced oncogenesis.33
Agonistic/ Xenobiotic/ Pharmacologic Second Hit
One prominent feature of chronic alcohol intake is its ability to sensitize the liver for agonistic activation of pathogen-associated pattern recognition receptors such as toll-like receptors (TLRs). The most classical demonstration of this effect is induction of massive liver necrosis and inflammation by injection of the TLR4 ligand, LPS in ethanol-fed animals.36 Indeed, the path-ogenetic roles of gut-derived endotoxin, LPS binding protein, CD14, and Kupffer cell activation in experimental ALD, have been convincingly demonstrated by a series of studies with gut sterilization, Kupffer cell depletion, and genetic mice performed by Ron Thurman’s laboratory.37 Alcohol intake increases messenger ribonucleic acid (mRNA) expression of not only TLR4, but also TLR2 and TLR6, which are activated by peptidoglycan; TLR7 and TLR8, which recognize viral single-stranded RNA; and TLR9, which recognizes bacterial and viral unmethylated CpG-containing DNA.38 Accordingly, administration of the ligands to some of these receptors, particularly for TLR2/6 and TLR4, aggravates alcoholic liver injury in mice.38 Interestingly, this study also demonstrates synergistic liver damage in ethanol-fed mice with polyIC and flagellin, the ligands for TLR3 and TLR4, despite the fact that mRNA levels of these receptors and TNFα are not increased by alcohol feeding or/and ligand treatment.38 Alcohol also primes T cells and this priming results in concavalin A-induced hepatitis in alcohol-fed rats; this pathology is reproduced in control mice after adaptive transfer of T cells from alcohol-fed rats.39 This model deserves further investigation for mechanisms of liver T cell priming by alcohol feeding. Alcohol-induced adaptive immunity can also be a target for the second hit. This is exemplified by experimental hepatitis and liver fibrosis induced by administration of acetaldehyde adducts in ethanol-fed guinea pigs40,41 or proinflammatory and profibrogenic responses elicited by malondialdehyde-acetaldehyde adducts (MAA), more naturally occurring hybrid adducts in ALD.42,43
Induction of CYP2E1 or isozymes as discussed above in relation to HFD, can be achieved by xenobiotics such as carbon tetrachloride or pyrazole, which can serve as a second hit to promote alcohol or LPS-induced liver cell death and inflammation44 and to even induce liver fibrosis.45,46 Increased susceptibility of alcoholic patients to acetaminophen toxicity has long been recognized.47 This interaction may be due to induction of CYP2E1 by alcohol with more robust production of N-acetyl-p-quinoneimine, a toxic metabolite of acetaminophen, or more susceptible hepatic mitochondria with reduced glutathione in alcohol-consuming subjects, which are readily targeted by acetaminophen-mediated cell death signaling involving c-Jun N-terminal kinasen (JNK).48 A second hit model with acetaminophen has been reproduced in rats given repetitive alcohol binge intake.49
The hormones, epinephrine, and thyroxin can also render a second hit insult to aggravate experimental ALD in rodents.50,51 These manipulations have the scientific rationale that alcohol consumption results in hypermetabolic state of the liver mediated by catecholamines52 or thyroid hormones,53 which contributes to accentuated centrilobular hypoxia and injury in ALD. The effects of catecholamine may also cause induction of plasminogen activator inhibitor-1 (PAI-1), which favors procoagulative and proinflammatory conditions in experimental ALD54 as seen in patients with ALD.55
Hemodynamic Second Hit
As discussed above, chronic alcoholism results in increased hepatic oxygen consumption and consequent centrilobular hypoxia due to the hypermetabolic state and inadequate compensation by increased oxygen delivery via portal blood flow.56 Such liver is vulnerable to ischemia/reperfusion due to accentuated oxidant stress, NFκB activation, chemokine expression, and neutrophilic infiltration,57 or transient hypoxia because of adenosine triphosphate (ATP) depletion.58 The procoagulative condition, with induced PAI-1 as discussed above, may also contribute to microcirculation disturbance at the sinusoidal level and worsen hypoxic insult.
HCV Proteins as Second Hits
Compelling evidence exists for synergistic liver damage caused by alcohol and HCV, which culminates in an increased incidence of HCC. Recent studies with mice expressing HCV proteins have shed pivotal insights into the mechanisms underlying the synergism. The HCV core protein causes overproduction of reactive oxygen species, which appears to be responsible for mitochondrial DNA damage.59 The core protein also inhibits microsomal triglyceride transfer protein activity and very low density lipoprotein secretion,60 which may underlie the genesis of fatty liver. The core protein also induces IR in mice and cell lines. This effect may be mediated by degradation of insulin receptor substrates (IRSs) 1 and 2 via upregulation of SOCS3,61 in a manner dependent on PA28γ,62 or via IRS serine phosphorylation.63 Thus, these core-induced perturbations such as oxidant stress and IR, which are known risk factors for ALD, can explain the synergism reproduced in alcohol-fed core transgenic mice.64 A most recent study with mice that express one of the HCV’s nonstructural proteins, NS5A in a hepatocyte-specific manner, further demonstrates synergistic alcoholic liver damage and liver oncogenesis with this viral protein. These consequences are attributable to NS5A-induced TLR4, the pattern recognition receptor known to be indispensable for experimental ALD.65 Second hit models utilizing these genetic mouse models are expected to shed more molecular insights into the most important and clinically relevant pathologic consequences of synergism resulting from alcohol and HCV.
Genetic Second Hit
Genetic second hit models basically apply either a gain of function approach for a suspected causal gene such as CYP2E166 or a loss of function manipulation for a protective gene such as Nrf2, superoxide dismutase 1 (CuZnSOD), interleukin-6 (IL-6), or cystathionine β synthase (CBS)67–70 as examples. The former is intended to promote an injurious mechanism, and the latter approach sensitizes the liver for alcohol-mediated harmful effects. The best example of a genetic model that does not require any genetic manipulation is that of female sex.71 Using the intragastric ethanol infusion model, female rats were shown to exhibit more accelerated liver damage with more panlobular hepatic steatosis, heightened inflammation, and plasma aminotransferase levels.71 Why the female gender causes an increased susceptibility to ALD is an important and clinically relevant question. Based on studies with female animals, several potential mechanisms have been proposed to date. These include less gastric, first-pass metabolism,72 increased endotoxemia,73 higher responsiveness of Kupffer cells to endotoxin,74 more lysosomal leakage and less BHMT expression,75 and enhanced osteopontin expression.76
Nrf2 (NF-E2-related factor 2) is a basic leucine zipper transcription factor that binds to the antioxidant-responsive element to induce transcription of detoxification and antioxidant genes such as glutathione S-transferase, NAD(P)H:quinine oxidoreductase-1, glutamate-cysteine ligase, heme oxygenase-1, thioredoxin reductase 1, and glucose-6-phosphate dehydrogenase. Nrf2 deficiency aggravates acetaminophen hepatotoxicity77 and alcoholic liver injury.67 Oxidant stress and protein kinases activate cytoplasmic Nrf2 by disrupting Nrf2-Keap1 complex leading to nuclear translocation. Interestingly, PKR-like ER kinase (PERK), one of the ER stress effector kinases, also phosphorylates and activates Nrf2 to facilitate a survival response.78 Thus, oxidant stress and ER stress converge onto Nrf2, and regulation and/or polymorphism of this gene may underlie a different predisposition to ALD. Deficiency in CuZnSOD expectedly results in increased sensitivity to alcoholic liver injury primarily characterized by oxidant damage.68 Mice deficient in IL-6 are also susceptible to alcohol-induced apoptosis and steatosis of the liver,69 potentially resulting from insufficient STAT activation.79 To this end, it is intriguing that there appears to be cell-type dependent roles of STAT in alcoholic liver injury such that STAT3 in hepatocytes promotes inflammation, whereas STAT3 in macrophages and Kupffer cells suppresses inflammation,79 highlighting the importance of cell-type specific research for the genetic second hit manipulation. Leptin deficiency results in sensitization for alcoholic steatohepatitis, which was attributed to suppressed metallothionein expression.7 However, different aspects of metabolic complications associated with obesity and NAFLD, may also be involved in this heightened sensitivity. Whether naturally occurring obesity and IR synergistically enhance alcoholic liver injury, needs to be determined as the models such as fa/fa or ob/ob or that based on choline and methionine deficiency, are not relevant to the background commonly seen in obese patients or patients with diabetes. As discussed above, genetic manipulation of any gene involved in the transmethylation or transsulfuration pathways, will be of interest for the second hit analysis. To this end, a preliminary study on cystathionine β synthase (CBS) heterozygosity demonstrates enhanced alcoholic liver pathology and a marked reduction in SAMe/SAH ratio.70 The latter effect should severely impair methyltransferase activities and DNA methylation.
FUTURE DIRECTIONS FOR IDEAL ALD ANIMAL MODELS
Despite many decades of rigorous attempts, the field still suffers from a lack of animal models that reproduce the whole spectrum of ALD. However, this in facts tells us the essence of the ALD pathogenesis – the complexity of gene–environment interactions that underlie the pathogenesis. One environmental factor that we know is critical is excessive intake of alcohol. Even though a threshold dose may differ depending on the existence of genetic and environmental risk factors, the fact is that patients consume at least 40 g/day for a minimum of 10 years. This has to be incorporated as a basic requirement when testing “second hit” in animal models if maximal recapitulation of human ALD pathology is desired.
The second requirement is that “second hit” has to be natural and best reflect patient conditions. Knocking out one gene or excessive pharmacologic treatments may not be natural, but hemizygosity or reproducing natural lifestyle environments may be more conducive to development of better two hit models. These more “subtle” or natural hits should be combined with “excessive” alcohol intake to test “multiple hits” in the genesis of ALD.
Lastly, we need to pay more attention to comorbidities. Patients with severe end-stage ALD who require liver transplantation, often have comorbidities such as viral hepatitis, obesity, and diabetes. Animal models reproducing synergistic pathological outcome caused by these comorbidities, are urgently needed for mechanistic research and identification of therapeutic targets.
ACKNOWLEDGMENTS
Preparation of this manuscript was supported by the National Institute on Alcohol Abuse and Alcoholism grants P50AA11999, R21AA016682, R24AA12885, and VA Merit Review and Senior Career Scientist awards. The authors also thank the excellent administrative support by Rosy Macias and Rebecca Handan.
ABBREVIATIONS
- ALD
alcoholic liver disease
- ALT
alanine aminotransferase
- ASH
alcoholic steatohepatitis
- BHMT
betaine-homocysteine methyltransferase
- CBS
cystathionine β synthase
- CuZnSOD
copper zinc superoxide dismutase
- CYP2E1
cytochrome P450 2E1
- ER
endoplasmic reticulum
- GNMT
glycine N-methyltransferase
- HCC
hepatocellular carcinoma
- HCV
hepatitis C virus
- HFD
high fat diet
- IKK
IkappaB kinase
- IL
interleukin
- IR
insulin resistance
- IRS
insulin receptor substrate
- Jak
Janus kinases
- JNK
c-Jun N-terminal kinase
- LPS
lipopolysaccharide
- MAA
malondialdehyde-acetaldehyde adducts
- mRNA
messenger ribonucleic acid
- NAFLD
nonalcoholic fatty liver disease
- NASH
nonalcoholic steatohepatitis
- NFkB
nuclear factor kappa B
- PAI-1
plasminogen activator inhibitor-1
- PERK
PKR-like ER kinase
- PPAR
peroxisome proliferator-activated receptor
- SAH
S-adenosylhomocysteine
- SAMe
S-adenosylmethionine
- SOCS
suppressor of cytokine signaling
- SREBP-1c
sterol regulatory element binding protein-1c
- STAT
signal transducer and activator of transcription
- TLR
toll-like receptor
- TNFα
tumor necrosis factor-α;
REFERENCES
- 1.Younossi ZM. Epidemiology of alcohol-induced liver disease. In: McCullough AJ, editor. Clinics in Liver Disease. Philadelphia: WB Saunders; 1998. pp. 661–671. [Google Scholar]
- 2.Day CP, James OF. Steatohepatitis: a tale of two “hits”? Gastroenterology. 1998;114(4):842–845. doi: 10.1016/s0016-5085(98)70599-2. [DOI] [PubMed] [Google Scholar]
- 3.Yoo JS, Ning SM, Pantuck CB, Pantuck EJ, Yang CS. Regulation of hepatic microsomal cytochrome P450IIE1 level by dietary lipids and carbohydrates in rats. J Nutr. 1991;121(7):959–965. doi: 10.1093/jn/121.7.959. [DOI] [PubMed] [Google Scholar]
- 4.Tsukamoto H, Towner SJ, Ciofalo LM, French SW. Ethanol-induced liver fibrosis in rats fed high fat diet. Hepatology. 1986;6(5):814–822. doi: 10.1002/hep.1840060503. [DOI] [PubMed] [Google Scholar]
- 5.Tsukada H, Wang PY, Kaneko T, Wang Y, Nakano M, Sato A. Dietary carbohydrate intake plays an important role in preventing alcoholic fatty liver in the rat. J Hepatol. 1998;29(5):715–724. doi: 10.1016/s0168-8278(98)80251-1. [DOI] [PubMed] [Google Scholar]
- 6.Korourian S, Hakkak R, Ronis MJ, et al. Diet and risk of ethanol-induced hepatotoxicity: carbohydrate-fat relationships in rats. Toxicol Sci. 1999;47(1):110–117. doi: 10.1093/toxsci/47.1.110. [DOI] [PubMed] [Google Scholar]
- 7.Tomita K, Azuma T, Kitamura N, et al. Leptin deficiency enhances sensitivity of rats to alcoholic steatohepatitis through suppression of metallothionein. Am J Physiol Gastrointest Liver Physiol. 2004;287(5):G1078–G1085. doi: 10.1152/ajpgi.00107.2004. [DOI] [PubMed] [Google Scholar]
- 8.Schattenberg JM, Wang Y, Singh R, Rigoli RM, Czaja MJ. Hepatocyte CYP2E1 overexpression and steatohepatitis lead to impaired hepatic insulin signaling. J Biol Chem. 2005;280(11):9887–9894. doi: 10.1074/jbc.M410310200. [DOI] [PubMed] [Google Scholar]
- 9.Ozcan U, Cao Q, Yilmaz E, et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science. 2004;306(5695):457–461. doi: 10.1126/science.1103160. [DOI] [PubMed] [Google Scholar]
- 10.Kim DS, Jeong SK, Kim HR, Kim DS, Chae SW, Chae HJ. Effects of triglyceride on ER stress and insulin resistance. Biochem Biophys Res Commun. 2007;363(1):140–145. doi: 10.1016/j.bbrc.2007.08.151. [DOI] [PubMed] [Google Scholar]
- 11.Kaplowitz N, Than TA, Shinohara M, Ji C. Endoplasmic reticulum stress and liver injury. Semin Liver Dis. 2007;27(4):367–377. doi: 10.1055/s-2007-991513. [DOI] [PubMed] [Google Scholar]
- 12.Nanji AA, Khwaja S, Tahan SR, Sadrzadeh SM. Plasma levels of a novel noncyclooxygenase-derived prostanoid (8-isoprostane) correlate with severity of liver injury in experimental alcoholic liver disease. J Pharmacol Exp Ther. 1994;269(3):1280–1285. [PubMed] [Google Scholar]
- 13.Tipoe GL, Liong EC, Casey CA, et al. A voluntary oral ethanol-feeding rat model associated with necroinflammatory liver injury. Alcohol Clin Exp Res. 2008;32(4):669–682. doi: 10.1111/j.1530-0277.2008.00623.x. [DOI] [PubMed] [Google Scholar]
- 14.Larter CZ, Yeh MM, Cheng J, et al. Activation of peroxisome proliferator-activated receptor alpha by dietary fish oil attenuates steatosis, but does not prevent experimental steatohepatitis because of hepatic lipoperoxide accumulation. J Gastroenterol Hepatol. 2008;23(2):267–275. doi: 10.1111/j.1440-1746.2007.05157.x. [DOI] [PubMed] [Google Scholar]
- 15.Wada S, Yamazaki T, Kawano Y, Miura S, Ezaki O. Fish oil fed prior to ethanol administration prevents acute ethanol-induced fatty liver in mice. J Hepatol. 2008;49(3):441–450. doi: 10.1016/j.jhep.2008.04.026. [DOI] [PubMed] [Google Scholar]
- 16.Powell LW. The role of alcoholism in hepatic iron storage disease. Ann N Y Acad Sci. 1975;252:124–134. doi: 10.1111/j.1749-6632.1975.tb19149.x. [DOI] [PubMed] [Google Scholar]
- 17.Sadrzadeh SM, Nanji AA, Price PL. The oral iron chelator, 1,2-dimethyl-3-hydroxypyrid-4-one reduces hepatic-free iron, lipid peroxidation and fat accumulation in chronically ethanol-fed rats. J Pharmacol Exp Ther. 1994;269(2):632–636. [PubMed] [Google Scholar]
- 18.Lin M, Rippe RA, Niemela O, Brittenham G, Tsukamoto H. Role of iron in NF-kappa B activation and cytokine gene expression by rat hepatic macrophages. Am J Physiol. 1997;272(6 Pt 1):G1355–G1364. doi: 10.1152/ajpgi.1997.272.6.G1355. [DOI] [PubMed] [Google Scholar]
- 19.Tsukamoto H, Horne W, Kamimura S, et al. Experimental liver cirrhosis induced by alcohol and iron. J Clin Invest. 1995;96(1):620–630. doi: 10.1172/JCI118077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tsukamoto H, Lin M, Ohata M, Giulivi C, French SW, Brittenham G. Iron primes hepatic macrophages for NF-kappaB activation in alcoholic liver injury. Am J Physiol. 1999;277(6 Pt 1):G1240–G1250. doi: 10.1152/ajpgi.1999.277.6.G1240. [DOI] [PubMed] [Google Scholar]
- 21.Otogawa K, Kinoshita K, Fujii H, et al. Erythrophagocytosis by liver macrophages (Kupffer cells) promotes oxidative stress, inflammation, and fibrosis in a rabbit model of steatohepatitis: implications for the pathogenesis of human nonalcoholic steatohepatitis. Am J Pathol. 2007;170(3):967–980. doi: 10.2353/ajpath.2007.060441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xiong S, She H, Zhang AS, et al. Hepatic macrophage iron aggravates experimental alcoholic steatohepatitis. Am J Physiol Gastrointest Liver Physiol. 2008;295(3):G512–G521. doi: 10.1152/ajpgi.90327.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McClain CJ, Antonow DR, Cohen DA, Shedlofsky SI. Zinc metabolism in alcoholic liver disease. Alcohol Clin Exp Res. 1986;10(6):582–589. doi: 10.1111/j.1530-0277.1986.tb05149.x. [DOI] [PubMed] [Google Scholar]
- 24.Cravo ML, Gloria LM, Selhub J, et al. Hyperhomocysteinemia in chronic alcoholism: correlation with folate, vitamin B-12, and vitamin B-6 status. Am J Clin Nutr. 1996;63(2):220–224. doi: 10.1093/ajcn/63.2.220. [DOI] [PubMed] [Google Scholar]
- 25.Barak AJ, Tuma DJ, Sorrell MF. Relationship of ethanol to choline metabolism in the liver: a review. Am J Clin Nutr. 1973;26(11):1234–1241. doi: 10.1093/ajcn/26.11.1234. [DOI] [PubMed] [Google Scholar]
- 26.Halsted CH, Villanueva JA, Devlin AM, et al. Folate deficiency disturbs hepatic methionine metabolism and promotes liver injury in the ethanol-fed micropig. Proc Natl Acad Sci U S A. 2002;99(15):10072–10077. doi: 10.1073/pnas.112336399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nieto N, Rojkind M. Repeated whiskey binges promote liver injury in rats fed a choline-deficient diet. J Hepatol. 2007;46(2):330–339. doi: 10.1016/j.jhep.2006.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lu SC, Tsukamoto H, Mato JM. Role of abnormal methionine metabolism in alcoholic liver injury. Alcohol. 2002;27(3):155–162. doi: 10.1016/s0741-8329(02)00226-4. [DOI] [PubMed] [Google Scholar]
- 29.Barak AJ, Beckenhauer HC, Tuma DJ. Methionine synthase. a possible prime site of the ethanolic lesion in liver. Alcohol. 2002;26(2):65–67. doi: 10.1016/s0741-8329(01)00201-4. [DOI] [PubMed] [Google Scholar]
- 30.Barak AJ, Beckenhauer HC, Junnila M, Tuma DJ. Dietary betaine promotes generation of hepatic S-adenosylmethio-nine and protects the liver from ethanol-induced fatty infiltration. Alcohol Clin Exp Res. 1993;17(3):552–555. doi: 10.1111/j.1530-0277.1993.tb00798.x. [DOI] [PubMed] [Google Scholar]
- 31.Ji C, Kaplowitz N. Betaine decreases hyperhomocysteinemia, endoplasmic reticulum stress, and liver injury in alcohol-fed mice. Gastroenterology. 2003;124(5):1488–1499. doi: 10.1016/s0016-5085(03)00276-2. [DOI] [PubMed] [Google Scholar]
- 32.Ji C, Shinohara M, Vance D, et al. Effect of transgenic extrahepatic expression of betaine-homocysteine methyl-transferase on alcohol or homocysteine-induced fatty liver. Alcohol Clin Exp Res. 2008;32(6):1049–1058. doi: 10.1111/j.1530-0277.2008.00666.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Martinez-Chantar ML, Vazquez-Chantada M, Ariz U, et al. Loss of the glycine N-methyltransferase gene leads to steatosis and hepatocellular carcinoma in mice. Hepatology. 2008;47(4):1191–1199. doi: 10.1002/hep.22159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Avila MA, Berasain C, Torres L, et al. Reduced mRNA abundance of the main enzymes involved in methionine metabolism in human liver cirrhosis and hepatocellular carcinoma. J Hepatol. 2000;33(6):907–914. doi: 10.1016/s0168-8278(00)80122-1. [DOI] [PubMed] [Google Scholar]
- 35.Tseng TL, Shih YP, Huang YC, et al. Genotypic and phenotypic characterization of a putative tumor susceptibility gene, GNMT, in liver cancer. Cancer Res. 2003;63(3):647–654. [PubMed] [Google Scholar]
- 36.Bhagwandeen BS, Apte M, Manwarring L, Dickeson J. Endotoxin induced hepatic necrosis in rats on an alcohol diet. J Pathol. 1987;152(1):47–53. doi: 10.1002/path.1711520107. [DOI] [PubMed] [Google Scholar]
- 37.Wheeler MD, Kono H, Yin M, et al. The role of Kupffer cell oxidant production in early ethanol-induced liver disease. Free Radic Biol Med. 2001;31(12):1544–1549. doi: 10.1016/s0891-5849(01)00748-1. [DOI] [PubMed] [Google Scholar]
- 38.Gustot T, Lemmers A, Moreno C, et al. Differential liver sensitization to toll-like receptor pathways in mice with alcoholic fatty liver. Hepatology. 2006;43(5):989–1000. doi: 10.1002/hep.21138. [DOI] [PubMed] [Google Scholar]
- 39.Cao Q, Batey R, Pang G, Clancy R. Ethanol-altered liver-associated T cells mediate liver injury in rats administered Concanavalin A (Con A) or lipopolysaccharide (LPS) Alcohol Clin Exp Res. 1999;23(10):1660–1667. [PubMed] [Google Scholar]
- 40.Yokoyama H, Ishii H, Nagata S, Kato S, Kamegaya K, Tsuchiya M. Experimental hepatitis induced by ethanol after immunization with acetaldehyde adducts. Hepatology. 1993;17(1):14–19. [PubMed] [Google Scholar]
- 41.Yokoyama H, Nagata S, Moriya S, et al. Hepatic fibrosis produced in guinea pigs by chronic ethanol administration and immunization with acetaldehyde adducts. Hepatology. 1995;21(5):1438–1442. [PubMed] [Google Scholar]
- 42.Tuma DJ. Role of malondialdehyde-acetaldehyde adducts in liver injury. Free Radic Biol Med. 2002;32(4):303–308. doi: 10.1016/s0891-5849(01)00742-0. [DOI] [PubMed] [Google Scholar]
- 43.Thiele GM, Duryee MJ, Freeman TL, et al. Rat sinusoidal liver endothelial cells (SECs) produce pro-fibrotic factors in response to adducts formed from the metabolites of ethanol. Biochem Pharmacol. 2005;70(11):1593–1600. doi: 10.1016/j.bcp.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 44.Lu Y, Cederbaum AI. Enhancement by pyrazole of lipopolysaccharide-induced liver injury in mice: role of cytochrome P450 2E1 and 2A5. Hepatology. 2006;44(1):263–274. doi: 10.1002/hep.21241. [DOI] [PubMed] [Google Scholar]
- 45.Bosma A, Brouwer A, Seifert WF, Knook DL. Synergism between ethanol and carbon tetrachloride in the generation of liver fibrosis. J Pathol. 1988;156(1):15–21. doi: 10.1002/path.1711560106. [DOI] [PubMed] [Google Scholar]
- 46.Hall PD, Plummer JL, Ilsley AH, Cousins MJ. Hepatic fibrosis and cirrhosis after chronic administration of alcohol and “low-dose” carbon tetrachloride vapor in the rat. Hepatology. 1991;13(5):815–819. doi: 10.1016/0270-9139(91)90246-r. [DOI] [PubMed] [Google Scholar]
- 47.Maddrey WC. Hepatic effects of acetaminophen. Enhanced toxicity in alcoholics. J Clin Gastroenterol. 1987;9(2):180–185. doi: 10.1097/00004836-198704000-00014. [DOI] [PubMed] [Google Scholar]
- 48.Hanawa N, Shinohara M, Saberi B, Gaarde WA, Han D, Kaplowitz N. Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen-induced liver injury. J Biol Chem. 2008;283(20):13565–13577. doi: 10.1074/jbc.M708916200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.McCuskey RS, Bethea NW, Wong J, et al. Ethanol binging exacerbates sinusoidal endothelial and parenchymal injury elicited by acetaminophen. J Hepatol. 2005;42(3):371–377. doi: 10.1016/j.jhep.2004.11.033. [DOI] [PubMed] [Google Scholar]
- 50.von MC, Beier JI, Guo L, Kaiser JP, Arteel GE. Contribution of the sympathetic hormone epinephrine to the sensitizing effect of ethanol on LPS-induced liver damage in mice. Am J Physiol Gastrointest Liver Physiol. 2008;294(5):G1227–G1234. doi: 10.1152/ajpgi.00050.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li J, French BA, Fu P, French SW. Liver necrosis induced by thyroid hormone administration in rats fed ethanol. Exp Mol Pathol. 2001;71(1):79–88. doi: 10.1006/exmp.2001.2381. [DOI] [PubMed] [Google Scholar]
- 52.Yuki T, Thurman RG. The swift increase in alcohol metabolism. Time course for the increase in hepatic oxygen uptake and the involvement of glycolysis. Biochem J. 1980;186(1):119–126. doi: 10.1042/bj1860119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Israel Y, Kalant H, Orrego H, Khanna JM, Videla L, Phillips JM. Experimental alcohol-induced hepatic necrosis: suppression by propylthiouracil. Proc Natl Acad Sci USA. 1975;72(3):1137–1141. doi: 10.1073/pnas.72.3.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bergheim I, Guo L, Davis MA, et al. Metformin prevents alcohol-induced liver injury in the mouse: critical role of plasminogen activator inhibitor-1. Gastroenterology. 2006;130(7):2099–2112. doi: 10.1053/j.gastro.2006.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Seth D, Hogg PJ, Gorrell MD, McCaughan GW, Haber PS. Direct effects of alcohol on hepatic fibrinolytic balance: implications for alcoholic liver disease. J Hepatol. 2008;48(4):614–627. doi: 10.1016/j.jhep.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 56.Tsukamoto H, Xi XP. Incomplete compensation of enhanced hepatic oxygen consumption in rats with alcoholic centrilobular liver necrosis. Hepatology. 1989;9(2):302–306. doi: 10.1002/hep.1840090223. [DOI] [PubMed] [Google Scholar]
- 57.Yamada S, Iida T, Tabata T, et al. Alcoholic fatty liver differentially induces a neutrophil-chemokine and hepatic necrosis after ischemia-reperfusion in rat. Hepatology. 2000;32(2):278–288. doi: 10.1053/jhep.2000.9604. [DOI] [PubMed] [Google Scholar]
- 58.French SW, Benson NC, Sun PS. Centrilobular liver necrosis induced by hypoxia in chronic ethanol-fed rats. Hepatology. 1984;4(5):912–917. doi: 10.1002/hep.1840040521. [DOI] [PubMed] [Google Scholar]
- 59.Moriya K, Nakagawa K, Santa T, et al. Oxidative stress in the absence of inflammation in a mouse model for hepatitis C virus-associated hepatocarcinogenesis. Cancer Res. 2001;61(11):4365–4370. [PubMed] [Google Scholar]
- 60.Perlemuter G, Sabile A, Letteron P, et al. Hepatitis C virus core protein inhibits microsomal triglyceride transfer protein activity and very low density lipoprotein secretion: a model of viral-related steatosis. FASEB J. 2002;16(2):185–194. doi: 10.1096/fj.01-0396com. [DOI] [PubMed] [Google Scholar]
- 61.Kawaguchi T, Yoshida T, Harada M, et al. Hepatitis C virus down-regulates insulin receptor substrates 1 and 2 through up-regulation of suppressor of cytokine signaling 3. Am J Pathol. 2004;165(5):1499–1508. doi: 10.1016/S0002-9440(10)63408-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Miyamoto H, Moriishi K, Moriya K, et al. Involvement of the PA28gamma-dependent pathway in insulin resistance induced by hepatitis C virus core protein. J Virol. 2007;81(4):1727–1735. doi: 10.1128/JVI.01683-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Banerjee S, Saito K, Ait-Goughoulte M, Meyer K, Ray RB, Ray R. Hepatitis C virus core protein upregulates serine phosphorylation of insulin receptor substrate-1 and impairs the downstream akt/protein kinase B signaling pathway for insulin resistance. J Virol. 2008;82(6):2606–2612. doi: 10.1128/JVI.01672-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Koike K, Tsutsumi T, Miyoshi H, et al. Molecular basis for the synergy between alcohol and hepatitis C virus in hepatocarcinogenesis. J Gastroenterol Hepatol. 2008;23(Suppl 1):S87–S91. doi: 10.1111/j.1440-1746.2007.05292.x. [DOI] [PubMed] [Google Scholar]
- 65.Machida K, Tsukamoto H, Mkrtchyan H, et al. Toll-like receptor 4 mediates synergism between alcohol and HCV in hepatic oncogenesis involving stem cell marker Nanog. Proc Natl Acad Sci USA. 2009;106(5):1548–1553. doi: 10.1073/pnas.0807390106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Morgan K, French SW, Morgan TR. Production of a cytochrome P450 2E1 transgenic mouse and initial evaluation of alcoholic liver damage. Hepatology. 2002;36(1):122–134. doi: 10.1053/jhep.2002.33720. [DOI] [PubMed] [Google Scholar]
- 67.Lamle J, Marhenke S, Borlak J, et al. Nuclear factor-eythroid 2-related factor 2 prevents alcohol-induced fulminant liver injury. Gastroenterology. 2008;134(4):1159–1168. doi: 10.1053/j.gastro.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 68.Kessova IG, Ho YS, Thung S, Cederbaum AI. Alcohol-induced liver injury in mice lacking Cu, Zn-superoxide dismutase. Hepatology. 2003;38(5):1136–1145. doi: 10.1053/jhep.2003.50450. [DOI] [PubMed] [Google Scholar]
- 69.El-Assal O, Hong F, Kim WH, Radaeva S, Gao B. IL-6-deficient mice are susceptible to ethanol-induced hepatic steatosis: IL-6 protects against ethanol-induced oxidative stress and mitochondrial permeability transition in the liver. Cell Mol Immunol. 2004;1(3):205–211. [PubMed] [Google Scholar]
- 70.Esfandiari F, Medici V, Wong DH, et al. Methyl regulation of ethanol induced steatosis in CBS deficient mouse. Alcohol Clin Exp Res. 2008:343A. [Google Scholar]
- 71.Iimuro Y, Frankenberg MV, Arteel GE, Bradford BU, Wall CA, Thurman RG. Female rats exhibit greater susceptibility to early alcohol-induced liver injury than males. Am J Physiol. 1997;272(5 Pt 1):G1186–G1194. doi: 10.1152/ajpgi.1997.272.5.G1186. [DOI] [PubMed] [Google Scholar]
- 72.Baraona E, Abittan CS, Dohmen K, et al. Gender differences in pharmacokinetics of alcohol. Alcohol Clin Exp Res. 2001;25(4):502–507. [PubMed] [Google Scholar]
- 73.Kono H, Wheeler MD, Rusyn I, et al. Gender differences in early alcohol-induced liver injury: role of CD14, NF-kappaB, and TNF-alpha. Am J Physiol Gastrointest Liver Physiol. 2000;278(4):G652–G661. doi: 10.1152/ajpgi.2000.278.4.G652. [DOI] [PubMed] [Google Scholar]
- 74.Ikejima K, Enomoto N, Iimuro Y, et al. Estrogen increases sensitivity of hepatic Kupffer cells to endotoxin. Am J Physiol. 1998;274(4 Pt 1):G669–G676. doi: 10.1152/ajpgi.1998.274.4.G669. [DOI] [PubMed] [Google Scholar]
- 75.Donohue TM, Curry-McCoy TV, Nanji AA, et al. Lysosomal leakage and lack of adaptation of hepatoprotective enzyme contribute to enhanced susceptibility to ethanol-induced liver injury in female rats. Alcohol Clin Exp Res. 2007;31(11):1944–1952. doi: 10.1111/j.1530-0277.2007.00512.x. [DOI] [PubMed] [Google Scholar]
- 76.Banerjee A, Apte UM, Smith R, Ramaiah SK. Higher neutrophil infiltration mediated by osteopontin is a likely contributing factor to the increased susceptibility of females to alcoholic liver disease. J Pathol. 2006;208(4):473–485. doi: 10.1002/path.1917. [DOI] [PubMed] [Google Scholar]
- 77.Chan K, Han XD, Kan YW. An important function of Nrf2 in combating oxidative stress: detoxification of acetaminophen. Proc Natl Acad Sci U S A. 2001;98(8):4611–4616. doi: 10.1073/pnas.081082098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Cullinan SB, Zhang D, Hannink M, Arvisais E, Kaufman RJ, Diehl JA. Nrf2 is a direct PERK substrate and effector of PERK-dependent cell survival. Mol Cell Biol. 2003;23(20):7198–7209. doi: 10.1128/MCB.23.20.7198-7209.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Horiguchi N, Wang L, Mukhopadhyay P, et al. Cell type-dependent pro- and anti-inflammatory role of signal transducer and activator of transcription 3 in alcoholic liver injury. Gastroenterology. 2008;134(4):1148–1158. doi: 10.1053/j.gastro.2008.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Takada A, Matsuda Y, Takase S. Effects of dietary fat on alcohol-pyrazole hepatitis in rats: the pathogenetic role of the nonalcohol dehydrogenase pathway in alcohol-induced hepatic cell injury. Alcohol Clin Exp Res. 1986;10(4):403–411. doi: 10.1111/j.1530-0277.1986.tb05114.x. [DOI] [PubMed] [Google Scholar]
- 81.Karaa A, Thompson KJ, McKillop IH, Clemens MG, Schrum LW. S-adenosyl-L-methionine attenuates oxidative stress and hepatic stellate cell activation in an ethanol-LPS-induced fibrotic rat model. Shock. 2008;30(2):197–205. doi: 10.1097/shk.0b013e318160f417. [DOI] [PubMed] [Google Scholar]