Abstract
Pluripotent stem cells hold significant promise in regenerative medicine due to their unlimited capacity for self-renewal, and potential to differentiate into any cell type of the body. In the current study, we demonstrate that proper mitochondrial function is essential for proliferation of undifferentiated embryonic stem cells (ESCs). Attenuating mitochondrial function under self-renewing conditions makes these cells more glycolytic-dependent, and is associated with an increase in the mRNA reserves of Nanog, Oct4 and Sox2. In contrast, attenuating mitochondrial function during the first 7 days of differentiation results in normal repression of Oct4, Nanog and Sox2. However differentiation potential is compromised as revealed by abnormal transcription of multiple Hox genes. Furthermore, under differentiating conditions in which mitochondrial function is attenuated, tumorigenic cells continue to persist. Our results, therefore establish the importance of normal mitochondrial function in ESC proliferation, regulating differentiation, and preventing the emergence of tumorigenic cells during the process of differentiation.
INTRODUCTION
The capacity of ESCs to undergo unlimited proliferation coupled with their ability to differentiate into diverse cell types makes them a powerful tool to study the biological mechanisms responsible for self-renewal, exit from pluripotent state and onset of lineage commitment. Current studies with ESCs are primarily focused on understanding the genetic regulation behind these processes. Similar attention has also been given to study the epigenetic mechanisms such as chromatin structure and the status of DNA methylation that contribute to the processes of self renewal and differentiation 1–3. Although these studies are extremely relevant and essential, very little is known about the relationship of cellular metabolism to self-renewal and early ESC differentiation 4.
In addition of being the “power house” of the cell, the mitochondrion plays a significant role in the metabolism of carbohydrates, lipids and proteins and acts as a hub for coordinating extrinsic and intrinsic signals to direct growth, proliferation differentiation and cell death 5, 6. It is well established that mitochondrial dysfunction has a major role in aging and apoptosis 7, 8, and that it contributes to a wide spectrum of diseases, including myopathies and neuropathies 9–11. However, relatively limited number of studies have been done to understand the importance of mitochondrial function in ESCs 4, 12–14. Recent studies using murine and human ESCs have revealed that under self-renewing conditions, ESCs typically have fewer mitochondria, which are arranged in perinuclear clusters that have poorly developed cristae with restricted oxidative capacity 13–16. Furthermore, both human and mouse ESCs express high levels of glycolytic enzymes and demonstrate high glycolytic flux 17, 18. Taken together, these results indicate that ESCs can function efficiently under anaerobic conditions and confirms recent reports demonstrating that hESCs exhibit improved self-renewal and pluripotency in 3–5% (physiological) O2 relative to atmospheric O2 19.
Upon differentiation, mitochondria in mouse and human ESC develop numerous cristae, increase in number and generate an extensive reticular network of tubular structures 13–17. This dramatic change in mitochondrial morphology is accompanied by higher rates of oxygen consumption, and increased production of ATP specifically through oxidative phosphorylation 15. Conversely, the expression of glycolytic enzymes and lactate production is significantly reduced compared to the self-renewing state 20. Collectively, these results have led to the hypothesis that mitochondrial oxidative phosphorylation is either dispensable, or only plays a minor role in ESC self-renewal, and is required only to meet the energy needs of differentiation.
In the present study, we addressed this hypothesis by attenuating mitochondrial function in (a) self-renewing and (b) differentiating mouse and human ESCs using a protonophore Carbonyl Cyanide m-Chlorophenylhydrazone (CCCP). Treating diverse types of cells, that include mammalian cell lines, yeast and even plant cells, with controlled doses of CCCP has been practiced extensively to knock-down mitochondrial function by chemical means21–24. Compared to other chemical inhibitors such as rotenone and oligomycin that affects specific subunits of the mitochondrial electron transport chain, CCCP treatment has a more robust effect on retarding mitochondrial function as it depolarizes the inner mitochondrial membrane to effectively uncouple oxidative phosphorylation from the electron transport chain. Our results clearly demonstrate that retarding mitochondrial activity have distinct effects in self-renewing ESCs and during early differentiation. While oxidative phosphorylation is required to support proliferation of self-renewing ESCs, the outcome of inhibiting oxidative phosphorylation during differentiation reveals the essential role of mitochondria in regulating proliferation and transcriptional activation of genes associated with early differentiation.
MATERIALS AND METHODS
Cells and Culture conditions
The hESC lines HSF-1 (UC01) and HSF-6 (UC06) were cultured and maintained on irradiated mouse embryonic fibroblast cell lines as described earlier25. The mESC line V6.5 was maintained on irradiated mouse embryonic fibroblasts in presence of LIF (Millipore). All work with hESCs was conducted with prior approval from the University of California Los Angeles Human Embryonic Stem Cell Research Oversight Committee. CCCP (carbonyl cyanide m-chloro phenyl hydrazone) (Sigma Aldrich), a chemical that uncouples oxidative phosphorylation from the electron transport chain was added to a final concentration of 2 μM to attenuate mitochondrial activity. This concentration of 2 μM was achieved after performing lethality analysis (LD50) with different concentrations of CCCP ranging from 1 μM to 12 μM. All the final concentrations of CCCP were prepared from a stock solution of 100mM in DMSO. Similar kind of solvent (DMSO) dilution was used for controls. For ATP and ROS assays self-renewing V6.5 and HSF1 cells were grown in absence of feeder cells on matrigel coated wells. Conditioned media (R&D Systems) was used to maintain the HSF1 cells in self-renewing state in a feeder free condition. The V6.5 cells were allowed to differentiate as an adherent culture in a feeder free condition with the withdrawal of LIF from the media. Differentiation of HSF1 cells was achieved by allowing them to grow as an adherent culture in absence of feeder cells and FGF2, and by replacing Knock-out (KO) Serum Replacement with Fetal Bovine Serum (FBS).
MitoTracker Red staining & Immunostaining
Live cells growing on cover-slips were incubated in the media containing MitoTracker Red (Molecular Probes) at a concentration of 250nM for 30 minutes, followed by three washes in the media and were fixed with 4% Formaldehyde in 1XPBS. The fixed cells were then permeabilized by treating with 0.2%TritonX 100 and immunostaining was performed following the protocol as described before 25. The primary Antibodies used were anti OCT3/4 (1:100; Santa Cruz Biotechnology Inc), NANOG (1:200; Abcam) and Secondary Antibodies (1:200) were from Jackson Immunoresearch Laboratories.
ATP and Reactive Oxygen Species (ROS) Assays
Intracellular ATP level was measured by the ATP Bioluminescence Assay Kit HS II (Roche) according to supplier’s protocol. The luminescence intensity was measured by using a Glomax 96 Microplate Luminometer (Promega) and the luminescence readings were normalized by cell count. For measurement of ROS levels, cells were incubated in a medium containing 2 μM dihydroethidium (Molecular Probes) at 37°C in dark for 15 minutes. Cells were then washed with 1XPBS buffer and suspended in 1XPBScontaining 0.5% BSA. The fluorescence intensity of 30,000 cells was recorded with the help of a BD Biosciences LSR II (BD Biosciences).
Cell death Assay
Apoptosis was evaluated using 7-aminoactinomycin D (7-AAD; Calbiochem) according to the manufacturers instructions followed by flow cytometry.
Teratoma
ESCs were grown in presence or absence of CCCP under feeder free conditions to determine the ability of tumor formation by self-renewing or differentiating cells. The self-renewing cells were grown for four days and harvested on day 4 for injection. For the differentiating ESCs, the cells were grown in differentiation media (absence of LIF) for seven days and then harvested. The cells were injected into the testes of SCID mice following the protocol as described previously 26. Surgery was performed following institutional approval for appropriate care and use of laboratory animals.
Analysis of Gene expression
RNA was extracted using the RNeasy Mini Kit (Qiagen), and reverse transcribed with Superscript II Reverse transcriptase (Invitrogen). Real time PCR was performed on the cDNA according to manufacturer’s protocol using Bio-Rad iQ iCycler with SYBR green as the fluorophore (Roche). For gene expression profiling of mouse and human stem cell samples, we used the Affymetrix Mouse Genome 430 2.0 Array and the Affymetrix human Genome U133plus2.0 array respectively. Two replicates were analyzed for each. Selection criteria for differential expression were performed as mentioned in the manuscript.
RESULTS
Dynamicity in mitochondrial morphology of self-renewing ESCs
Mitochondrial morphology was evaluated in self-renewing mouse and human ESCs by staining with a mitochondrial specific dye, Mitotracker Red. Consistent with previous reports, we found that both in self-renewing mouse (V6.5) and human (HSF1 and HSF6) ESCs mitochondria are distributed as perinuclear clusters that appear punctate and globular (Fig 1A–C″). Interestingly, similar mitochondrial morphology and distribution are observed even in human induced pluripotent cells (hiPS1; Fig. 1E, E′) derived by reprogramming dermal fibroblasts (Fig. 1D) with OCT4, SOX2, cMYC and KLF4 retroviruses 27 suggesting that epigenetic reprogramming to generate hIPS cell lines is also associated with mitochondrial reprogramming. Although the common punctate and rudimentary appearance of mitochondria are seen in all undifferentiated ESCs, the amount of mitochondrial mass varies between independent lines of hESCs. For instance HSF6 cells appear to have more mitochondria than HSF1 cells (compare Fig 1B and C). Interestingly, regardless of the cell line, a modest increase in mitochondrial content associated with mitochondrial fusion to generate distinct tubules is seen during mitosis (Fig. 1G, H, & K, L). Following cytokinesis, mitochondria once again reorganize as perinuclear clusters (Fig. 1I, M). These cell cycle phase dependent changes in mitochondrial morphology indicate that mitochondria might play a specific functional role under self-renewing conditions.
Figure 1. Dynamicity in mitochondrial morphology of self-renewing ESCs during cell division.

Mitochondrial morphology in mouse and human ESCs as evidenced by staining with mitochondrial specific dye Mitotracker Red (red). In all figures green shows expression of pluripotent transcription factor, OCT4 and the nuclei are stained with TO-PRO 3 (blue).
(A–C″) Punctate perinuclear mitochondria in self-renewing mESCs (V6.5) (A–A″) and in hESC lines HSF1 (B–B″) and HSF6 (C–C″).
(D) Reticulate mitochondrial network in human foreskin fibroblasts.
(E–E′) Induced pluripotent stem cells, hiPS1, reprogrammed from foreskin fibroblasts have rudimentary and punctate mitochondria, similar to that found in self-renewing hESCs (B–C″).
(F–M) Changes in mitochondrial mass and morphology in self-renewing HSF1 (F–I) and HSF6 (J–M) cells during different phases of mitotic division.
Proliferation of ESCs under self renewing conditions depend on mitochondrial activity
To investigate the importance of mitochondrial oxidative phosphorylation in ESC self-renewal, mitochondrial activity was perturbed in self-renewing mouse (V6.5) and human (HSF1) ESCs, growing in absence of feeder cells, by culturing them in presence of 2 μM CCCP (carbonyl cyanide m-chloro phenyl hydrazone). We found that this treatment resulted in a 30% drop in cellular ATP levels in mouse ESCs (Fig 2A), and a 3–4 fold increase in the level of cellular Reactive Oxygen species (ROS) (Fig. 2B). As shown in Fig 2C, this attenuation of mitochondrial function affects the proliferation rate of mouse ESCs. However, no change in cell cycle phasing of CCCP treated mouse ESCs was apparent when compared with the untreated controls (Fig. 2D) indicating that while proliferation rate is reduced, this is not associated with a block in specific steps in cell cycle progression. Importantly, upon staining with 7ADD, a marker for apoptotic cells, no difference in the number of 7ADD positive cells was observed between mouse ESCs growing in presence or absence of CCCP (Fig 2E). Furthermore, no difference in the proportion of senesced cells as determined by negative Ki67 staining was observed under self-renewing conditions (Fig. 2F–G′). Evaluation of the human ESC line, HSF1 maintained in the presence of 2 μM CCCP, revealed that in a manner similar to the mouse V6.5 cells these cells demonstrate changes in the cellular ATP (Fig. 2H) and ROS levels (Fig. 2I) and proliferate slowly (Fig. 2J). Collectively, these results clearly suggest that the proliferation of self-renewing ESCs is dependent on mitochondrial function.
Figure 2. Disrupting mitochondrial function in self-renewing ESCs significantly reduces proliferation rate.
(A) Normalized cellular ATP level in undifferentiated mouse ESCs (V6.5) treated with CCCP as measured by a luciferin-luciferase assay on the fourth day of treatment.
(B) Four days of CCP treatment lead to a significant increase in the cellular ROS level in CCCP treated undifferentiated mESCs compared to the untreated control cells.
(C) Proliferation rate of self-renewing V6.5 cells treated with CCCP (red) relative to untreated cells (black, control). After initial rounds of division, proliferation in CCCP treated cells get retarded.
(D) Distribution of cells (percentage of the total population) in different phases of cell cycle in untreated (blue) and CCCP treated self-renewing V6.5 cells (red).
(E) Percentage of cells undergoing apoptotic cell death as revealed by staining with 7ADD in CCCP treated (red) and untreated (blue) populations of self-renewing V6.5 ESCs plotted as a function of days.
(F–G′) Ki67 staining (red) of self-renewing V6.5 control cells (F, F′) or those treated with CCCP for four days (G, G′). The nuclei are marked with TO-PRO3 (blue).
(H) Cellular ATP level in CCCP treated self-renewing HSF1 cells, normalized to that observed in untreated control cells, on the fourth day of treatment.
(I) Increased cellular ROS level in CCCP treated HSF1 cells compared to untreated cells, four days after treatment.
(J) Proliferation profile of self-renewing HSF1 cells treated with CCCP (red) relative to untreated cells (black).
Pluripotency of self-renewing ESCs is not compromised upon blocking mitochondrial function
One of the fundamental properties of undifferentiated ESCs is to maintain a pluripotent state and therefore retain the capacity to generate endodermal, mesodermal, ectodermal and germ cell derivatives as they differentiate. In an attempt to understand the relationship between oxidative phosphorylation and pluripotency, we studied the mRNA levels of pluripotent transcription factors, OCT4, NANOG and SOX2 using real time RT-PCR in both mouse (Fig 3A) and human (Fig. 3B) ESCs grown in presence or absence of CCCP. We found that the expression of OCT4, NANOG and SOX2 transcripts were increased by at least two fold in both mouse and human ESCs treated with CCCP compared to their untreated controls. In order to evaluate the pluripotency of CCCP treated ESCs, we transplanted the treated and control ESCs into the testicles of adult SCID mice (n=4). Tumor formation was achieved in 100% of transplants from wild type and CCCP treated ESCs. Histological sections of the tumors revealed that CCCP treated ESCs generated teratomas, which were comparable to control, treated ESCs and showed evidence of differentiation into all three embryonic lineages (Fig. 3C–F).
Figure 3. Pluripotency is unaffected in undifferentiated ESCs with compromised mitochondrial function.
(A–B) Transcript levels of Oct4, Nanog and Sox2 as evidenced by real time RT-PCR in self-renewing mouse ESCs (A) and human ESCs (HSF1; B) treated with CCCP. For both V6.5 and HSF1 cells, the different colored dots represent the fold increase in expression upon CCCP treatment, normalized to untreated control cells (represented by a straight line =1). Results are from four independent experiments each performed in duplicate.
(C–F) Histological sections through the testis of SCID mice.
(C) Normal tissue architecture in histological section through the testis without any transplanted ESCs.
(D–F) Sections through testicular tumors derived from transplanting undifferentiated V6.5 cells (D) or CCCP treated V6.5 cells (E, F). EN: Endodermal derivative; ME: Mesodermal derivative; EC: Ectodermal derivative.
(G) Rate of glycolysis as measured by Extracellular Acidification Rate (ECAR) of CCCP treated and untreated self-renewing V6.5 cells.
(H) Oxygen Consumption rate (OCR) of the same sets of cells as shown in (G). Prior to time point (A) the cells were maintained at low Glucose condition and with addition of Glucose at time point (A), no variation in OCR is noticed in CCCP treated cells while a significant increase in OCR is observed in untreated cells (control). Administering 2deoxy Glucose at time point (B) lowers down the OCR in the control cells.
In order to confirm that CCCP treated mouse ESCs have attenuated mitochondrial function, we examined glycolytic flux (Fig. 3G) and the rate of oxygen consumption (Fig. 3H) in treated verses control ESCs cultured under self-renewing conditions. These results demonstrate that in the presence of CCCP, undifferentiated ESCs exhibit reduced consumption of oxygen due to the uncoupling of the electron transport chain and a concomitant increase in glycolytic flux. Taken together, these results demonstrate that blocking oxidative phosphorylation with CCCP in undifferentiated ESCs increases glycolytic metabolism and reduces proliferation rate which are associated with increased transcriptional of the three critical pluripotency transcription factors Oct4, Nanog and Sox2.
Differentiation of ESCs is associated with changes in mitochondrial morphology
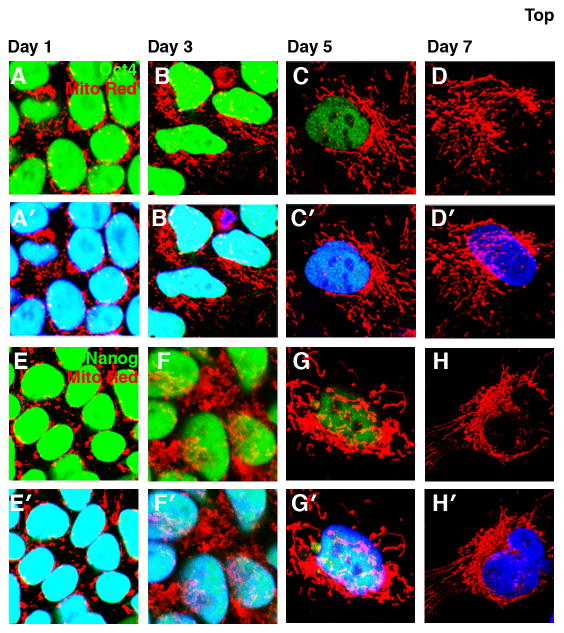
Previous studies have demonstrated that compared to the punctate globular mitochondria, as found in undifferentiated ESCs, differentiation results in the generation of an extensive network of branched mitochondria 13, 15. However correlating the kinetics of network formation with loss of pluripotent gene expression has never been performed. To achieve this, we evaluated the temporal changes in mitochondrial morphology with respect to the loss of OCT4 (Fig. 4A–D′) and NANOG (Fig. 4E–H′) in differentiating HSF1 cells. Differentiation was performed as an adherent culture in the absence of FGF2 and feeder cells, and by substituting Knock-out Serum Replacement with Fetal Bovine Serum. As shown in Fig. 4B, B′, the formation of mitochondrial network was initiated on day 3 of differentiation while the cells maintained high levels of OCT4 protein. On subsequent days of differentiation as OCT4 was lost, the cells acquired an extensive network of mitochondrial branching (Fig. 2C–D′). Identical temporal profile of mitochondrial branching was seen with respect to loss of NANOG expression in differentiating HSF1 cells (Fig. 4E–H′). Consistent with the formation of extensive mitochondrial network, differentiating HSF1 cells exhibited a gradual increase in cellular ATP and ROS levels. Our results, therefore, demonstrate that at a single cell level, mitochondrial branching precedes the loss of pluripotency markers and thereby suggests that establishing appropriate metabolic status of a cell is one of the earliest fate changes that occur when differentiation is initiated.
Figure 4. Initiation of mitochondrial branching during early differentiation precedes loss of NANOG and OCT4.
Mitotracker staining (red) of differentiating HSF1 cells on days 1, 3, 5 and 7 of differentiation in relation to the loss of OCT4 (A–D′) and NANOG (E–H′) as shown in green. The nuclei are marked with TO-PRO 3 (blue). Red and the green channels are shown in panel A–D and E-H, the merge of all three channels is shown in A′–D′ and E′–H′.
Proliferation and Early Differentiation is affected when differentiation is conducted under conditions that attenuate mitochondrial activity
To examine the importance of oxidative phosphorylation during early cell fate lineage commitment, we blocked oxidative phosphorylation in differentiating ESCs for seven days by growing them in presence of CCCP. This treatment dropped cellular ATP levels by 40% in both mouse and human ESCs (Fig. 5A). Furthermore, we determined that ROS levels were initially increased relative to control in the first few days after CCCP treatment, however, after seven days the level of ROS was normalized to wild type (data not shown) presumably due to the high levels of antioxidants produced by differentiating ESCs to keep ROS in balance 15. Staining the differentiated cells with Mitotracker Red revealed that, unlike controls, CCCP treated mouse and human differentiated ESCs exhibit an apparent discontinuous network (Fig. 5B–G), likely due to a block in mitochondrial fusion. Interestingly as observed for self-renewing ESCs, CCCP treatment affected the proliferation rate of differentiating mouse and human ESCs (Fig. 5H, I). Although the rate of proliferation were comparable for the first three days of differentiation, on subsequent days the CCCP treated differentiating cells demonstrated significantly reduced proliferation rates suggesting that oxidative phosphorylation is also required for proliferation during the process of differentiation.
Figure 5. Attenuated mitochondrial function represses proliferation in differentiating ESCs but does not affect loss of pluripotency.
(A) Cellular ATP levels in CCCP treated mESCs (blue) and hESCs (green), normalized to their corresponding untreated control cells at day 7 of differentiation.
(B–C) Mitotracker staining (red) of differentiating V6.5 cells in the absence (B) or presence (C) of CCCP for seven days. The nuclei are marked with TO-PRO3 (blue)
(D–G) Mitotracker staining (red) of differentiating HSF1 cells in the absence (D, F) or presence (E, G) of CCCP for seven days. A high magnification image of a portion of D and E, demonstrating the fragmentation of mitochondrial tubules is represented in F and G respectively.
(H, I) Proliferation profile of CCCP treated differentiating V6.5 (H) and HSF1 (I) cells (red). Compared to the untreated control cells (black), proliferation rate of CCCP treated cells gets retarded.
(J, K) Transcript levels of Oct4, Sox2 and Nanog in V6.5 cells (J) and HSF1 cells (K) differentiated in the presence of CCCP for seven days relative to undifferentiated cells and cells undergoing differentiation in absence of CCCP.
Differentiation of ESCs is characterized by the loss pluripotency, and a concomitant turning on of transcriptional programs required for differentiation. In an attempt to understand the contribution of mitochondrial activity in the process of differentiation we induced differentiation in the presence of CCCP. Our results show that CCCP treatment during differentiation had no effect on the repression of Oct4, Sox2 and Nanog expression in either mouse or human ESCs (Fig. 5J & K).
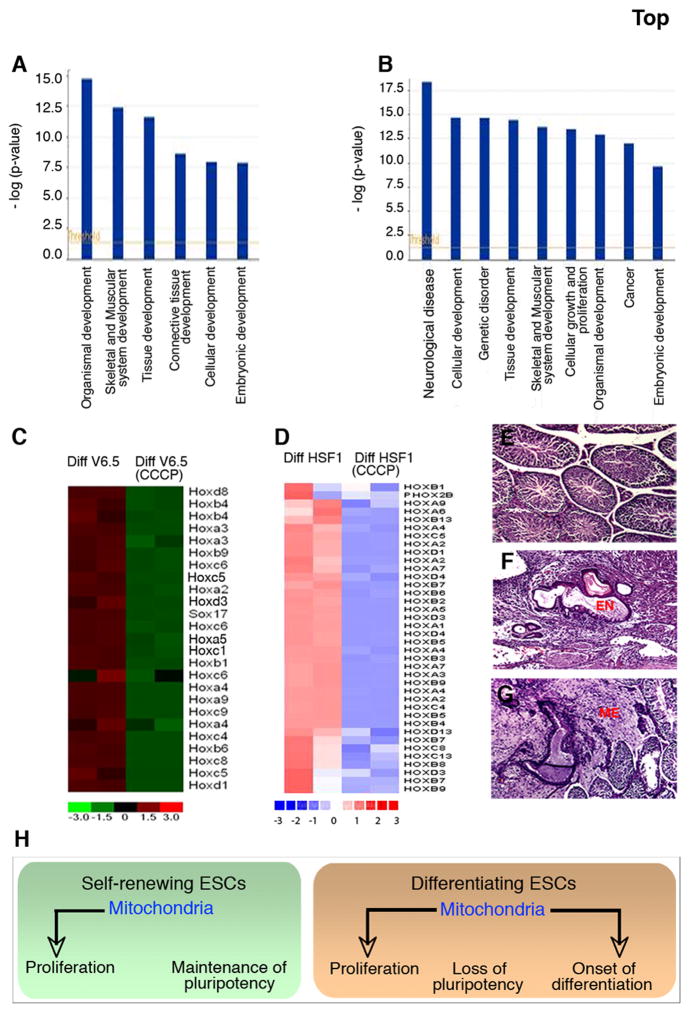
To identify transcriptional consequences of CCCP treatment during differentiation, we performed genome wide expression profiling comparing ESCs differentiated in CCCP for 7 or 10 days (mouse and human respectively) to that of untreated control cells. Using the GeneChip Mouse Genome 430 2.0 Array, we found 1200 genes differentially expressed by a factor of 2 or more on the basis of a t test p value of 0.001, in differentiated mouse ESCs treated with CCCP. Gene ontology (GO) based classification revealed that the differentially expressed genes are associated with early development and differentiation (Fig. 6A), with a notable decrease in Hox gene expression (Fig. 6C) in differentiating mouse ESCs treated with CCCP. Using the Affymetrix human chip, U133plus2.0 array microarrays we compared differentiated human ESCs cultured in the presence or absence of CCCP. Applying the same set of criteria we found that 410 genes were differentially expressed between control and CCCP treated differentiating hESCs cells. GO analysis revealed that CCCP treated differentiating hESCs also exhibited differentially expressed genes associated with development and differentiation (Fig. 6B). Furthermore, in a manner similar to mouse ESCs, differentiated human ESCs demonstrated a significant decrease in the expression of HOX genes when differentiated in the presence of CCCP (Fig. 6D). Together, these results clearly demonstrate that in differentiating ESCs normal mitochondrial activity is critical for the onset of differentiation.
Figure 6. Attenuated mitochondrial function results in compromised differentiation.
(A, B) Gene ontology based clustering of genes that are differentially expressed in differentiating V6.5 (A) and HSF1 (B) cells treated with CCCP compared to the untreated controls at day 7 of differentiation.
(C, D) Expression profile of Hox genes in CCCP treated differentiating V6.5 (C) and HSF1 (D) cells relative to their respective untreated control cells at day 7 of differentiation. Gene expression profiling was performed in two independent replicates. The guide at the bottom of the figure indicates the fold difference of expression between the samples with green or blue indicating lower expression and red indicating higher expression.
(E–G) Histological sections through the testis of SCID mice.
(E) Transplantation of V6.5 cells on day seven of differentiation does not result in tumor formation.
(F, G) In contrast, introduction of differentiated V6.5 cells cultured in the presence of CCCP for 7 days results in the formation of teratomas. EN: Endoderm, ME: Mesoderm, EC: Ectoderm.
(H) A model for mitochondrial control of different processes in self-renewing and early differentiating ESCs.
Differentiating ESCs attenuated for mitochondrial activity cause teratoma
Given that repression of Oct4, Nanog and Sox2 mRNA were unchanged relative to controls, but execution of transcriptional programs associated with differentiation were compromised in CCCP treated differentiating ESCs, we next evaluated whether differentiating in the presence of CCCP promotes the persistence of cells that are still capable of tumor formation. To achieve this we transplanted V6.5 cells differentiated in the presence or absence of CCCP for seven days, into the testicles of adult SCID mice. Transplantation of untreated differentiated cells resulted in zero tumor formation 6 weeks after transplant26. In contrast, cells differentiated in the presence of CCCP resulted in tumor formation in 100% of transplants (n=4). Histological analysis of these tumors showed teratoma formation with differentiation into cell types representing the three embryonic lineages (Fig. 6E–G). These results indicate that CCCP treatment during the process of differentiation promotes the persistence of cells with tumorigenic potential when transplanted into SCID mice.
DISCUSSION
This study describes the role of mitochondrial activity in regulating proliferation of both self-renewing and differentiating ESCs and also highlights the link between normal mitochondrial function and the onset of differentiation program during early lineage specification of ESCs (Fig. 6H). We demonstrate that attenuating mitochondrial function, as evidenced by a drop in cellular ATP level and a concomitant increase in the level of ROS, retards the proliferation rate of self-renewing mouse and human ESCs without compromising their pluripotency. Interestingly, no block in cell cycle progression is induced; rather a proportional slow down of all the phases of cell cycle is observed. Similar kind of response has been previously reported in Drosophila cells containing a mutation in the gene encoding mitochondrial ribosomal protein, mRpl12 28. Importantly, even with this drop in ATP level, significant metabolic activities are still retained such that these cells do not apoptose or undergo senescence. Although the proliferation of self-renewing ESCs is slower, their pluripotency is maintained, given the transcriptional load of the key pluripotency genes Oct4, Nanog and Sox2 are elevated at least two fold in CCCP treated murine and human ESCs. We speculate that this increase in expression of the pluripotent markers is likely to be due to a metabolic shift as the CCCP treated self-renewing ESCs were found to be more glycolytic dependent. In this connection it is interesting to note that previous studies have demonstrated that normally self-renewing ESCs express higher levels of glycolytic genes compared to that in differentiated cells 18. Here we demonstrate that upon blocking mitochondrial function while the proliferation of self-renewing ESCs is retarded, these cells become more glycolytic dependent and express pluripotent markers at a level even higher than their normal counterparts. Together, these results indicate an interesting dichotomy in that self-renewing ESCs may require mitochondrial function for proliferation, while glycolysis supports the regulation of the pluripotency network co-regulated by Oct4, Nanog and Sox2.
Previous studies had shown that ESCs cultured under self-renewing conditions are characterized by the presence of punctate and globular mitochondria, together with limited oxygen consumption and reduced levels of ATP. In the current study we first evaluated mitochondrial morphology in fibroblasts and IPS cells to determine whether these particular mitochondrial features were restricted to pluripotent stem cells derived from the inner cell mass of human and mouse blastocysts, or can be attributed to human pluripotent stem cells of non-embryonic origin. Our results demonstrate that epigenetic reprogramming of dermal fibroblasts with OCT4, SOX2, cMYC and KLF4 retroviruses also causes mitochondrial reprogramming from a branched reticular network to punctuate organelles that are equivalent in morphology to what is routinely identified in hESCs. This result suggests that punctuate mitochondrial morphology and presumably reduced oxidative phosphorylation are required to maintain the pluripotent state.
In contrast, differentiated ESCs have an extensive network of branched mitochondria and a concomitant increase in oxygen consumption. However, prior to the current analysis, the kinetics of this network formation relative to the loss of pluripotency was not well established. Here we demonstrate that the induction of mitochondrial branching precedes loss of NANOG and OCT4 protein, indicating that establishing the appropriate metabolic status of a cell is one of the earliest fate changes upon induction of differentiation. Previous studies in Drosophila as well as in other systems have demonstrated that mitochondria exert specific effects on cellular function through mechanisms collectively defined as retrograde signaling 29–31. It is possible that in differentiating ESCs, as the cells lose their pluripotency, specific retrograde signals from the mitochondria to the nucleus is essential for the transcriptional triggering of the genes associated with early differentiation. Indeed, we found that disruption of mitochondrial function during ESC differentiation repressed transcriptional programs required for embryonic lineage differentiation, and in particular HOX gene expression both in mouse and human ESCs.
Hox genes encode DNA binding proteins that regulate gene expression and control various aspects of morphogenesis and early embryonic cell differentiation. Mammalian Hox genes are divided into two broad categories: a family of 38 genes that are related to the Antenapedia gene of Drosophila, and the other group is constituted of more distantly related genes. These 38 genes, in turn, are organized into four clusters (Hox clusters) each containing 8–11 genes. We found that majority of genes transcribed from all four Hox gene clusters are repressed in mouse as well as human ESCs differentiating under inhibited mitochondrial activity. Interestingly, although the execution of transcriptional programs associated with differentiation were abnormal, normal repression of the transcription factors required to support self renewal was observed. Therefore, in order to determine whether this uncoupling of pluripotent gene repression from the normal execution of differentiation increased the risk of tumorigenicity, we transplanted the differentiated cells into the testicles of SCID mice. We found that differentiated cells with attenuated mitochondrial function developed tumors in 100% of transplants, with the tumor type being diagnosed as a teratoma. Whether this is due to small numbers of pluripotent cells remaining in the population of mitochondrially challenged cells or whether differentiating cells with lower mitochondrial function can revert back to pluripotency in vivo remains to be addressed.
In this context it is important to note that the chromatin in ESCs has been found to be highly enriched with a specific modification pattern termed as “bivalent domain.” These domains consist of large regions of H3 lysine 27 methylation harboring smaller regions of H3 lysine 4 methylation and thereby combine both repressive and activating modifications. Interestingly, they are associated with genes that play important roles in embryonic development and lineage specification. It has been hypothesized that in ES cells the bivalent domains silence developmental genes while preserving their potential to get activated upon initiation of specific differentiation programs 32. This raises the possibility that blocking mitochondrial function in differentiating ESCs might lead to stable epigenetic modifications in these bivalent domains that prevent further activation of the genes associated with them, even after the effect of CCCP is released as the cells are injected into testis. From a therapeutic point of view, such distinction may be irrelevent since these results argue for discarding any ES line with metabolic challenge much as one sets standards for or against acceptable chromosomal and epigenetic changes in cell lines for clinical utility.
Acknowledgments
We thank William Lowry for providing human IPS cells and for many helpful discussions. We thank Michael A. Teitell for joint funding support and discussions. We thank Owen Witte for valuable advice, Xinmin Li for help with microarray analysis and Karen Reue for access to the XF24 Extracellular Flux Analyzer. This work was supported by a seed grant from California Institute of Regenerative Medicine (CIRM) and an innovator award from The Broad Stem Cell Research Center (BSCRC).
Footnotes
Author Contributions:
Sudip Mandal: Conception & Design; Collection and Assembly of Data, Data Analysis and Interpretation, Manuscript Writing and Final Approval of Manuscript.
Anne G. Lindgren: Conception & Design; Collection and Assembly of Data, Data Analysis and Interpretation.
Anand S. Srivastava: Assembly of Data, Data Analysis and Interpretation, and Manuscript writing.
Amander T. Clark: Conception & Design; Data Analysis and Interpretation, Manuscript writing, final approval of manuscript and advised and supervised the project.
Utpal Banerjee: Conception & Design; Data Analysis and Interpretation, Financial Support, Manuscript writing, final approval of manuscript and advised and supervised the project.
References
- 1.Chen L, Daley GQ. Molecular basis of pluripotency. Hum Mol Genet. 2008;17:R23–27. doi: 10.1093/hmg/ddn050. [DOI] [PubMed] [Google Scholar]
- 2.Gangaraju VK, Lin H. MicroRNAs: key regulators of stem cells. Nat Rev Mol Cell Biol. 2009;10:116–125. doi: 10.1038/nrm2621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ng JH, Heng JC, Loh YH, et al. Transcriptional and epigenetic regulations of embryonic stem cells. Mutat Res. 2008;647:52–58. doi: 10.1016/j.mrfmmm.2008.08.009. [DOI] [PubMed] [Google Scholar]
- 4.Bavister BD. The mitochondrial contribution to stem cell biology. Reprod Fertil Dev. 2006;18:829–838. doi: 10.1071/rd06111. [DOI] [PubMed] [Google Scholar]
- 5.McBride HM, Neuspiel M, Wasiak S. Mitochondria: more than just a powerhouse. Curr Biol. 2006;16:R551–560. doi: 10.1016/j.cub.2006.06.054. [DOI] [PubMed] [Google Scholar]
- 6.Newmeyer DD, Ferguson-Miller S. Mitochondria: releasing power for life and unleashing the machineries of death. Cell. 2003;112:481–490. doi: 10.1016/s0092-8674(03)00116-8. [DOI] [PubMed] [Google Scholar]
- 7.Suen DF, Norris KL, Youle RJ. Mitochondrial dynamics and apoptosis. Genes Dev. 2008;22:1577–1590. doi: 10.1101/gad.1658508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wallace DC. Mitochondrial genetics: a paradigm for aging and degenerative diseases? Science. 1992;256:628–632. doi: 10.1126/science.1533953. [DOI] [PubMed] [Google Scholar]
- 9.DiMauro S. Mitochondrial myopathies. Curr Opin Rheumatol. 2006;18:636–641. doi: 10.1097/01.bor.0000245729.17759.f2. [DOI] [PubMed] [Google Scholar]
- 10.DiMauro S, Schon EA. Mitochondrial disorders in the nervous system. Annu Rev Neurosci. 2008;31:91–123. doi: 10.1146/annurev.neuro.30.051606.094302. [DOI] [PubMed] [Google Scholar]
- 11.Yu-Wai-Man P, Griffiths PG, Hudson G, et al. Inherited mitochondrial optic neuropathies. J Med Genet. 2009;46:145–158. doi: 10.1136/jmg.2007.054270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Oh SK, Kim HS, Ahn HJ, et al. Derivation and characterization of new human embryonic stem cell lines: SNUhES1, SNUhES2, and SNUhES3. Stem Cells. 2005;23:211–219. doi: 10.1634/stemcells.2004-0122. [DOI] [PubMed] [Google Scholar]
- 13.St John JC, Amaral A, Bowles E, et al. The analysis of mitochondria and mitochondrial DNA in human embryonic stem cells. Methods Mol Biol. 2006;331:347–374. doi: 10.1385/1-59745-046-4:347. [DOI] [PubMed] [Google Scholar]
- 14.St John JC, Ramalho-Santos J, Gray HL, et al. The expression of mitochondrial DNA transcription factors during early cardiomyocyte in vitro differentiation from human embryonic stem cells. Cloning Stem Cells. 2005;7:141–153. doi: 10.1089/clo.2005.7.141. [DOI] [PubMed] [Google Scholar]
- 15.Cho YM, Kwon S, Pak YK, et al. Dynamic changes in mitochondrial biogenesis and antioxidant enzymes during the spontaneous differentiation of human embryonic stem cells. Biochem Biophys Res Commun. 2006;348:1472–1478. doi: 10.1016/j.bbrc.2006.08.020. [DOI] [PubMed] [Google Scholar]
- 16.Facucho-Oliveira JM, Alderson J, Spikings EC, et al. Mitochondrial DNA replication during differentiation of murine embryonic stem cells. J Cell Sci. 2007;120:4025–4034. doi: 10.1242/jcs.016972. [DOI] [PubMed] [Google Scholar]
- 17.Chung S, Dzeja PP, Faustino RS, et al. Mitochondrial oxidative metabolism is required for the cardiac differentiation of stem cells. Nat Clin Pract Cardiovasc Med. 2007;4 (Suppl 1):S60–67. doi: 10.1038/ncpcardio0766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kondoh H, Lleonart ME, Nakashima Y, et al. A high glycolytic flux supports the proliferative potential of murine embryonic stem cells. Antioxid Redox Signal. 2007;9:293–299. doi: 10.1089/ars.2006.1467. [DOI] [PubMed] [Google Scholar]
- 19.Ezashi T, Das P, Roberts RM. Low O2 tensions and the prevention of differentiation of hES cells. Proc Natl Acad Sci U S A. 2005;102:4783–4788. doi: 10.1073/pnas.0501283102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ramalho-Santos J, Varum S, Amaral S, et al. Mitochondrial functionality in reproduction: from gonads and gametes to embryos and embryonic stem cells. Hum Reprod Update. 2009;15:553–572. doi: 10.1093/humupd/dmp016. [DOI] [PubMed] [Google Scholar]
- 21.Griparic L, Kanazawa T, van der Bliek AM. Regulation of the mitochondrial dynamin-like protein Opa1 by proteolytic cleavage. J Cell Biol. 2007;178:757–764. doi: 10.1083/jcb.200704112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Handy DE, Lubos E, Yang Y, et al. Glutathione peroxidase-1 regulates mitochondrial function to modulate redox-dependent cellular responses. J Biol Chem. 2009;284:11913–11921. doi: 10.1074/jbc.M900392200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Holmuhamedov E, Lewis L, Bienengraeber M, et al. Suppression of human tumor cell proliferation through mitochondrial targeting. FASEB J. 2002;16:1010–1016. doi: 10.1096/fj.01-0996com. [DOI] [PubMed] [Google Scholar]
- 24.Rikhvanov EG, Gamburg KZ, Varakina NN, et al. Nuclear-mitochondrial cross-talk during heat shock in Arabidopsis cell culture. Plant J. 2007;52:763–778. doi: 10.1111/j.1365-313X.2007.03275.x. [DOI] [PubMed] [Google Scholar]
- 25.Park TS, Galic Z, Conway AE, et al. Derivation of primordial germ cells from human embryonic and induced pluripotent stem cells is significantly improved by coculture with human fetal gonadal cells. Stem Cells. 2009;27:783–795. doi: 10.1002/stem.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Conway AE, Lindgren A, Galic Z, et al. A self-renewal program controls the expansion of genetically unstable cancer stem cells in pluripotent stem cell-derived tumors. Stem Cells. 2009;27:18–28. doi: 10.1634/stemcells.2008-0529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lowry WE, Richter L, Yachechko R, et al. Generation of human induced pluripotent stem cells from dermal fibroblasts. Proc Natl Acad Sci U S A. 2008;105:2883–2888. doi: 10.1073/pnas.0711983105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Frei C, Galloni M, Hafen E, et al. The Drosophila mitochondrial ribosomal protein mRpL12 is required for Cyclin D/Cdk4-driven growth. EMBO J. 2005;24:623–634. doi: 10.1038/sj.emboj.7600523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Butow RA, Avadhani NG. Mitochondrial signaling: the retrograde response. Mol Cell. 2004;14:1–15. doi: 10.1016/s1097-2765(04)00179-0. [DOI] [PubMed] [Google Scholar]
- 30.Mandal S, Freije WA, Guptan P, et al. Metabolic control of G1-S transition: cyclin E degradation by p53-induced activation of the ubiquitin-proteasome system. J Cell Biol. 188:473–479. doi: 10.1083/jcb.200912024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mandal S, Guptan P, Owusu-Ansah E, et al. Mitochondrial regulation of cell cycle progression during development as revealed by the tenured mutation in Drosophila. Dev Cell. 2005;9:843–854. doi: 10.1016/j.devcel.2005.11.006. [DOI] [PubMed] [Google Scholar]
- 32.Bernstein BE, Mikkelsen TS, Xie X, et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell. 2006;125:315–326. doi: 10.1016/j.cell.2006.02.041. [DOI] [PubMed] [Google Scholar]